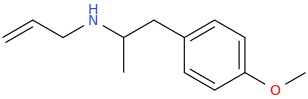
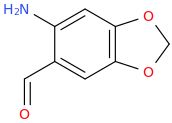
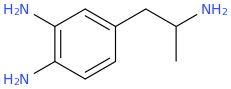
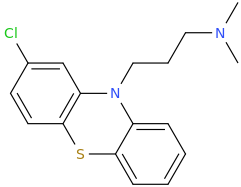
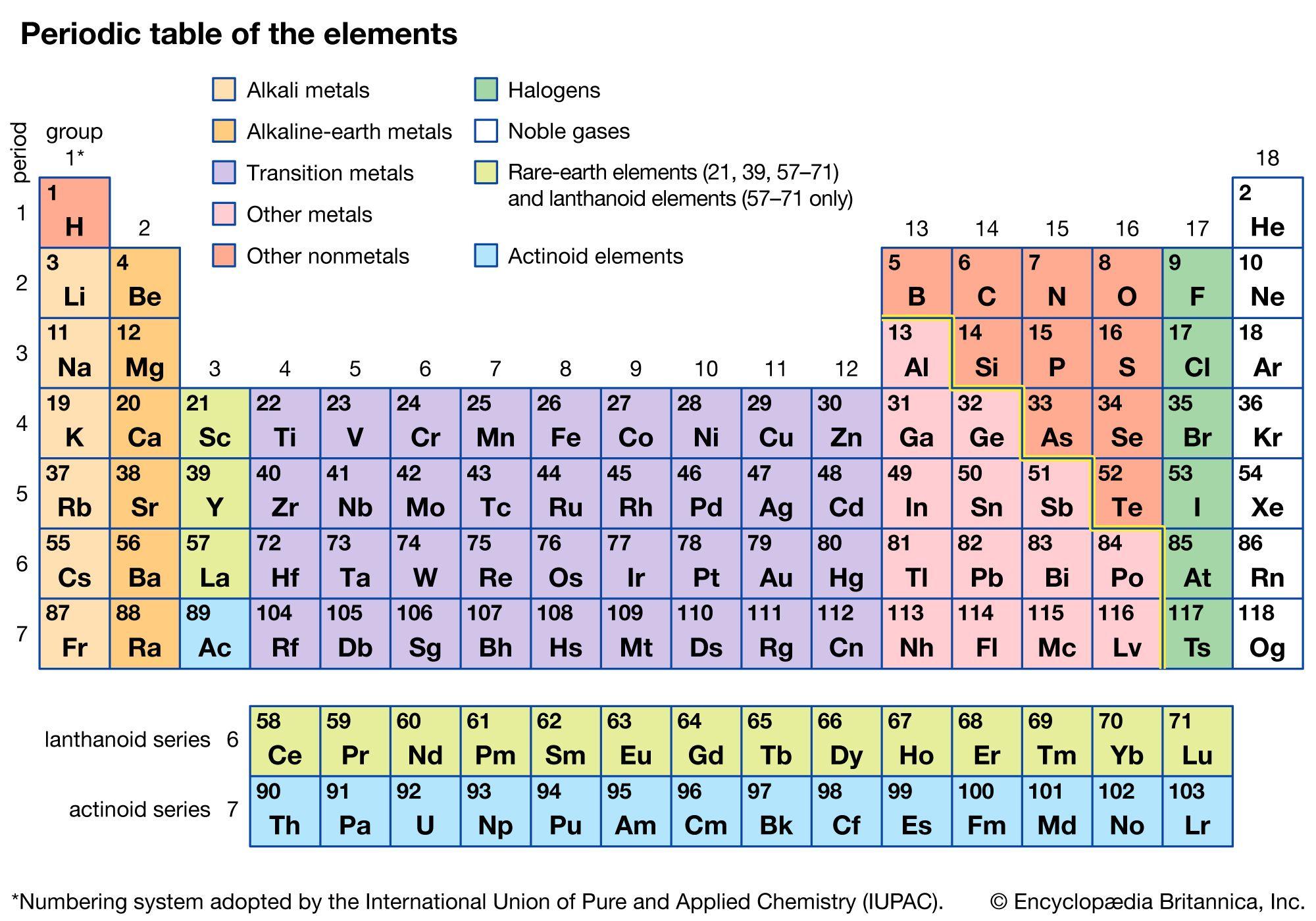
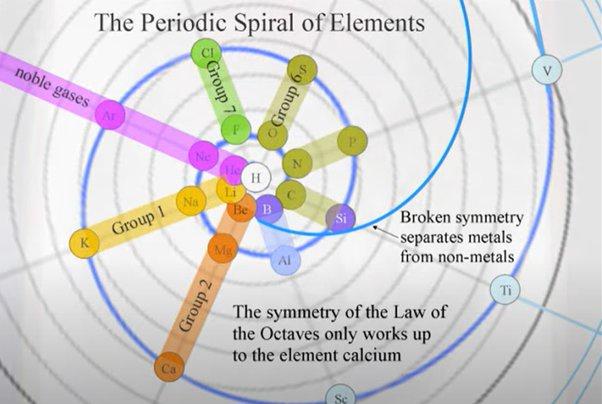
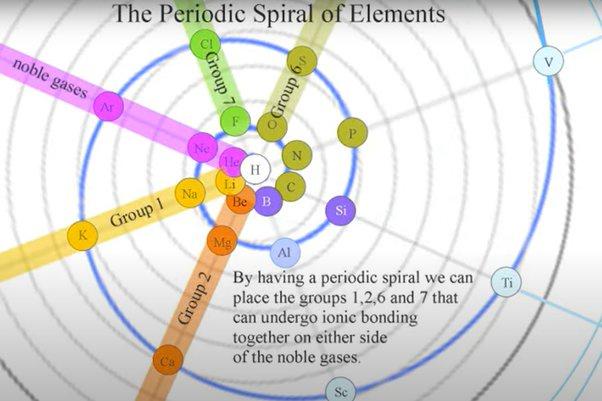
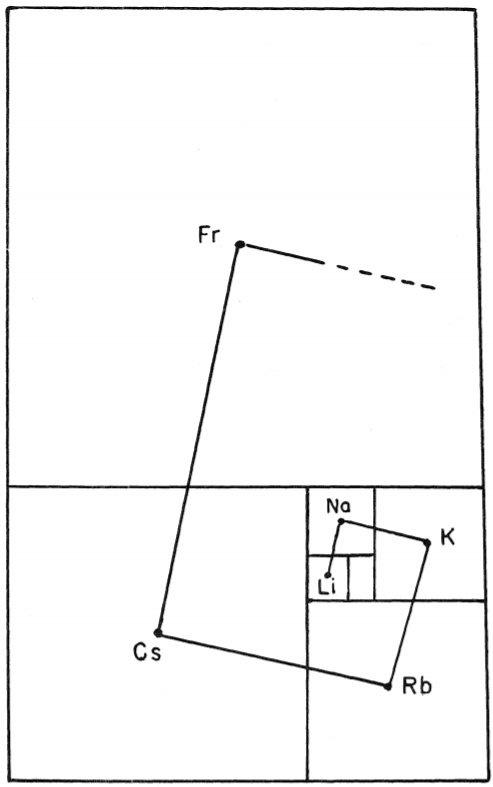
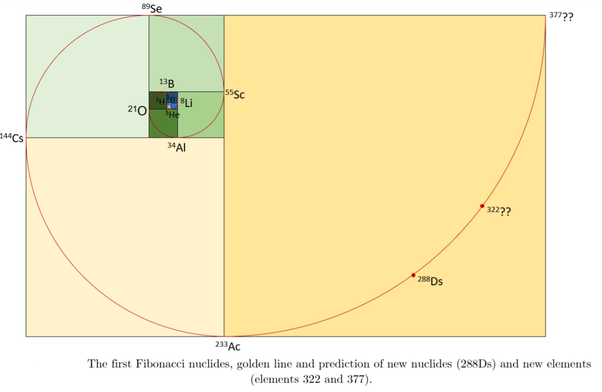
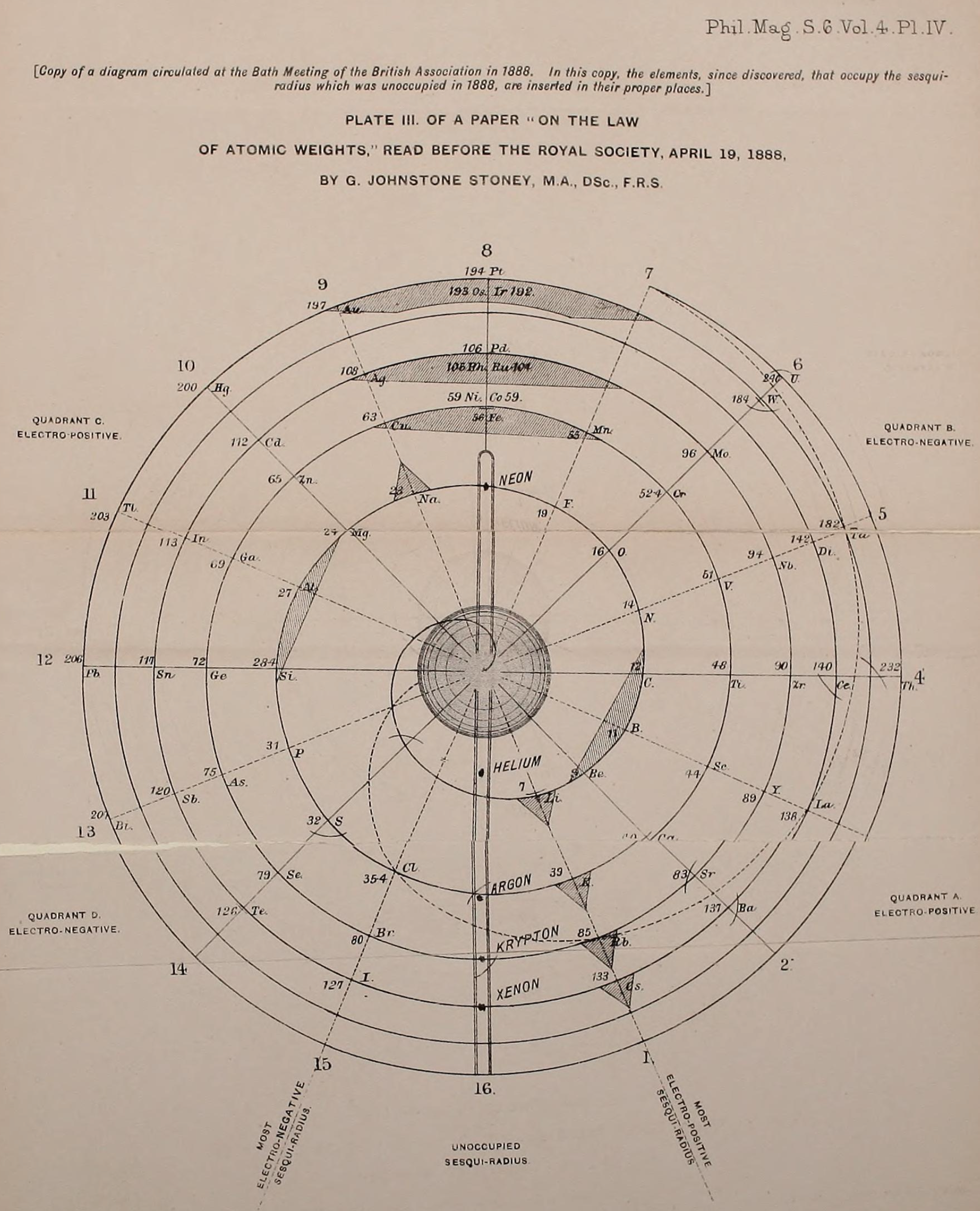
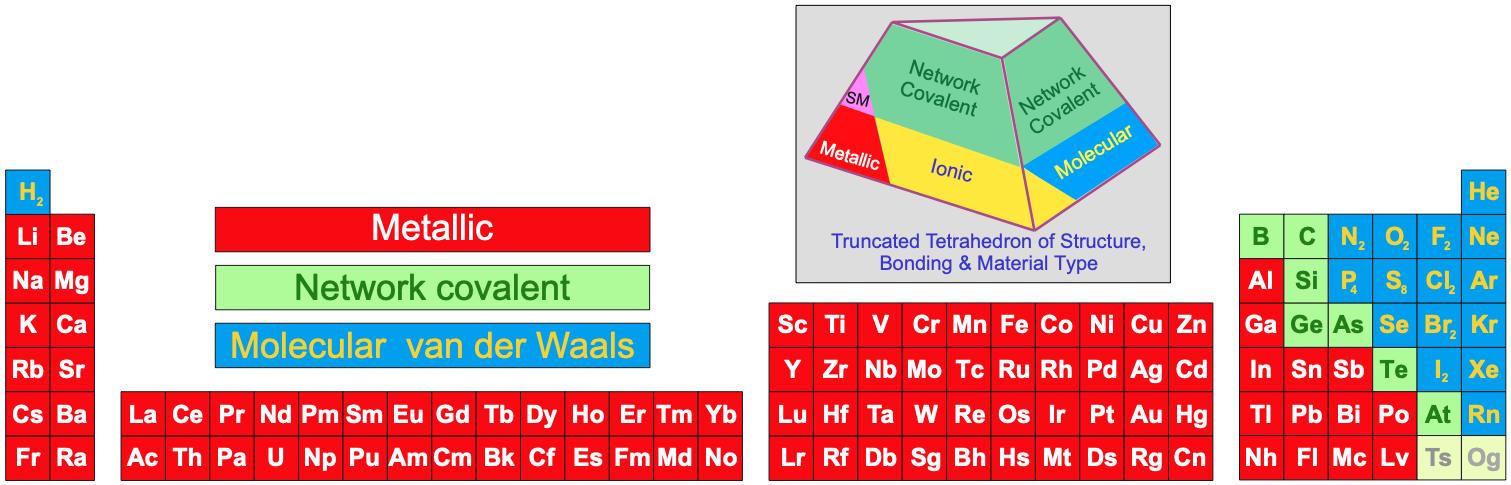
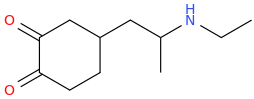
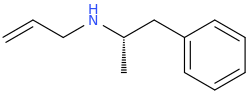
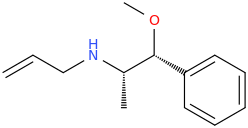
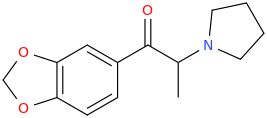
Been doing a lot of high end associative thinking lately about the way different STEM disciplines fit together. This morning I was thinking about music theory in relation to math in relation to chemistry (harmonics connects music to physics to chemistry since matter is vibrating) and it occurred to me that there should be a way to view the periodic table of elements in relation to the Fibonacci numbers.... Kind of relating back to the good old "rabbit problem" and ways to stack together recurring quantized shapes like particles. And sure enough after some googling -- there is! Tickled with myself that I was able to predict it. This is all the reading I've done on it so far but there's a very deep rabbit hole to go down here! Thought some of you guys may be interested.
Answer: Excellent question! I’m glad someone asked this here, as it’s a glaring example of the fact that all that happens in nature, is really only “symmetry-forming and breaking,” as some folks like to put it. If I may confess, even I was impressed by this theory at first, but soon realised tha...

chemistry1.quora.com
The article doesn't go into it, but part of what's going on is that the elements that fall "in between" the Fibonacci numbers tend to be more reactive, I believe, based on the number of electrons in their outer valence shell as one of the main trends. Which is part of why carbon, with it's four outermost electrons (half the full shell) is able to form the backbone of so much organic chemistry.
Recently had a couple DOC trips for the first time in over 8 years (have barely touched psychedelics since before my cancer) and it's got my brain firing on all gears! Spiral out my swirly friends!
Edit -- you know, "reactive" isn't necessarily the right term for it in general... More reactive as far as covalent bonding is concerned is perhaps a better way to look at the ones that fall in series with Carbon (that trend just on the organic side of the semimetal line). The ones that are further away from that series to be more ionically reactive, out to the point where they suddenly become nonreactive (noble gas series).
Images to go along with it for anyone that doesn't want to click the link. The visuals definitely help.
Notice how new groups in the standard periodic table tend to recur in a similar pattern as how the new color pairs recur in the classic depiction of the Fibonacci rabbit problem