-
Neuroscience & Pharmacology Discussion Welcome Guest
Posting Rules Bluelight Rules Recent Journal Articles Chemistry Mega-Thread FREE Chemistry Databases! Self-Education Guide -
N&PD Moderators: Skorpio
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.Ketamine salts solubility
- Thread starter fastandbulbous
- Start date
- Status
- Not open for further replies.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 10,235
How Many Times Have I Been Around?
Recycled Paper Is Paving The Ground.
Yes, She Lives For The Written Word.
And People Come Second Or Possibly Third. Then Page After Page Of Sniping Rage, From An English Poet Or An American Sage. So, Stop Me, O Ho Ho, Stop Me If You Think That You've Heard This One Before. I Still Love You. Yes, I Still Love You. Only Slightly, Only Slightly Less Than I Used To, My Love.Last edited:Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 10,235

TYLENOL (APAP)
4-hydroxy-N-acetylaniline
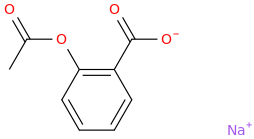
ASPIRIN
sodium 2-acetoxy-benzoate

ADVIL
sodium 2-(4-isobutyl-phenyl)propionate

GRAPE FLAVOR
2-aminobenzoate

RESVERATROL
(E)-1-(4-hydroxyphenyl)-2-(3,5-dihydroxyphenyl)ethene
2HC=CH2
ETHENE,
a plant Hormone
CH3CH2CH2CH2CH2-OH
AMYL ALCOHOLLast edited:Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 10,235
Fluorine Gets A Lot Of Hype For Being Electronegative, But It Lacks The Heavy Atom Effect Which Is Why Oxygen, The 2nd Most EN Element, Beats It Hands Down Pharmacologically.
Oxidation (loss of electrons) Is A Form Of Burning.
Oxygen Is Necessary To Burn Hydrocarbons But Cannot Itself Be Burned.
A Carbon With An Oxygen Attached Has Had Its Energy Lowered Thermodynamically.
Fluorine Is Most Useful If Making Teflon.
4-FA Is Just Aight.

AIGHT
1-(4-fluorophenyl)-2-aminopropane
Though F and O are both very electronegative, Halogens Are Deactivating (miotic) While O is activating (mydriatic).

DOCTOR WHO
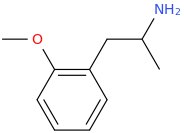
1-(4-methoxyphenyl)-2-aminopropane
I only sampled one batch of PMA, but it was so worth it, a pink Superman.
But what about all the youngsters who died from PMA, you shriek?! Chill out. You can die from anything.
The only person I condone Drug use for is myself, and if I petitioned the President for a Schedule I license, he just might grant my wish.Last edited:Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 10,235
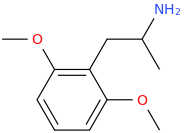
I Have Read That DOA (4-amino-2,5-dimethoxyamphetamine) is inactive, but I still like posting structures, even when their effects if any are unknown.
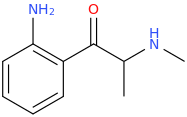
THE_GRAPE_APE
1-(2-aminophenyl)-1-oxo-2-methylaminopropane
^--Inspired by Grape flavoring, 2-aminobenzoate.

THE_MOTHER_LIQUOR
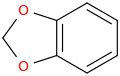
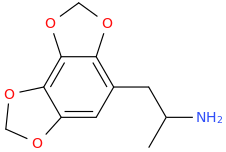
1-(2-amino-3,4-methylenedioxyphenyl)-2-aminopropane

SWEET_LIKE_LICORICE
1-(6-amino-3,4-methylenedioxyphenyl)-2-aminopropane
Dangerous Like Syphilis!Last edited:Fertile
Bluelighter
- Joined
- Mar 31, 2022
- Messages
- 1,627
Talaprevir, Telmisartan, Olmesartan, Losartan, Montelukast, Atorvastatin. What do all of these medications have in common? They violate Lipinski's Rules of 5. Now, why is that SO odd, well because Lipinski and his ego convinced Pfizer not to bother studying any drug that violated his RO5.
It's an excellent rule-of-thumb, but when finally Pfizer changed their opinion, 2 of the successful candidates had been in the Pfizer library for decades with no work carried out because of a rule.
I, for one, would trust it if I totally understood the receptor(s) and binding.... but with many unknowns, it's not possible to stay.
In fact I defeated RO5 myself. Only in a very basic way - by producing a mixed dimer based on the 7-ethynyl homologue of clobazam and the 6-ethynyl homologue of diazepam and a MW over 600, It remains a single chemical throughout ADME. Why? Because I realized and alcohol mimic needed a 1,4-benzo and a 1,5-benzo but it would cost twice as much (at least) to get a licence for 2 drugs--- thus they were joined with a butane-1,4-diol bridge between the 3 positions.
Some messing around with different 1,4 & 1,5-benzodiazepines will be needed but with a MW of 684, it's most certainly active.
It seems that be it a 1,4 or a 1,5, a 7-ethynyl (or 8-ethynyl if their is a 4th ring e.g. alprazolam) then the compounds ARE a5 selective, just like all of the positive effects of alcohol.
I MAY have to go with a bridge with an extra -OH or 2 to lower LogP but other than that, the bridge is known to work. What I REALL:Y seek is more information on etifoxine. I spent weeks looking and the ONLY thing I found was that a '2 chloro increases toxicity and reduces the effects.
I don't know if a '2 substitution of 1,5-benzos has any effects on activity.
8-ARYL-5,6,7,8-TETRAHY DROPYRAZOLO(3,4-b)(1,4)-DIAZEPINE-1H, 4H-5,7-DONES, AND MEDICAMENTSCONTAINING THESE
Rackur et al.
US 4305952A
It would seem so. Column 13 has a table and R6 represents substitution of the benzene ring. With the 1,4-benzodiazepines, '2 bstitution increased activity while 4 substitution produces a compound with peripheral activity so let's look at the '2 substitutions. While differing 2 & 4 substitutions are tried out, only the '2=-chloro & '2-methyl are tested. No logic of what was tested if given BUT...
Consider that methaqualone has a 2'methyl but it's close (I am told) relative is mecloqualone in which the methyl is swapper for a -Cl. At this stage, just an accident.
4,5-DIHYDRO-1,2,4-TRIAZOLO(4,3-A)QUINOXALIN-4-ONES
LOEV B; SUH J T; WILLIAMS B E; GEORGIEV V S
NZ197101A
Gives examples of 1,5-benzodiazepines and examples with triazolo rings. Of course, nothing to say they are highly active in man, but it's well worth knowing what is likely to be a worthwhile modification and what is not.
So anyone with time - I would appreciate someone checking my work.
It all adds up to the ability to more finely tune the final compound. OK, no doubt it will take many attempts, but if the triazolo works, the '2-subsstitution works and a 3-OH (for bridge) ALL work is well worth knowing.- Status
- Not open for further replies.