polymath
Bluelight Crew
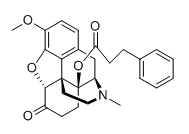
^ Quite unusual that this only has the N atom corresponding to the indole nitrogen... I tried to think of an ibogaine/harmala kind of derivative but you obviously can't build that from this compound.
N&PD Moderators: Skorpio | someguyontheinternet
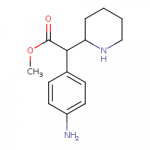

The 4-amino version of methylphenidate is significantly more potent than MPH itself, but can't know if it has aniline-like toxicity...
View attachment 17784
The SwissTargetPrediction also gives it a slim chance of binding to mu receptor.
If someone were to make a non-amine MPH with the piperidine nitrogen removed or replaced with oxygen, it would be easier to purify if it had an amino group on the benzene ring.
Edit1: Also, making it a 4-aminoethylphenidate gives it a little bit more likelihood of binding to mu receptor, similar to meperidine SAR.
Edit2: And, by reducing the shit out of antibiotic chloramphenicol, you could obtain an equivalent 4-amino version of amphetamine.
I've always mused about near methylphenidates that were also near fentanyls.
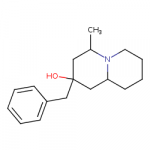
I drew some amine compounds like this in another thread:
View attachment 21584
It's a possible dopamine reuptake inhibitor and also looks a bit like fentanyl or meperidine, and a pharmacology app predicts it's a possible mu receptor ligand.
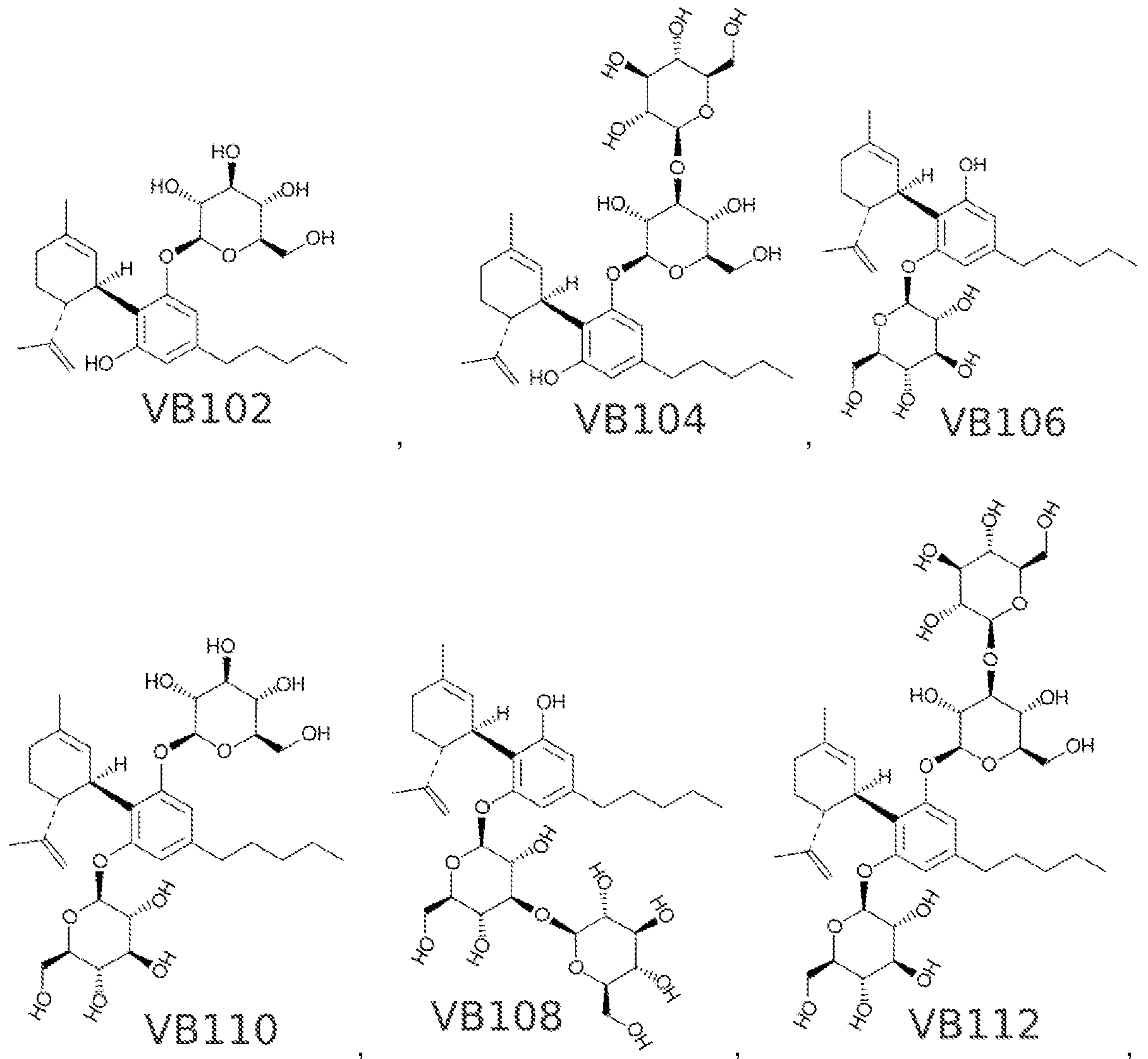
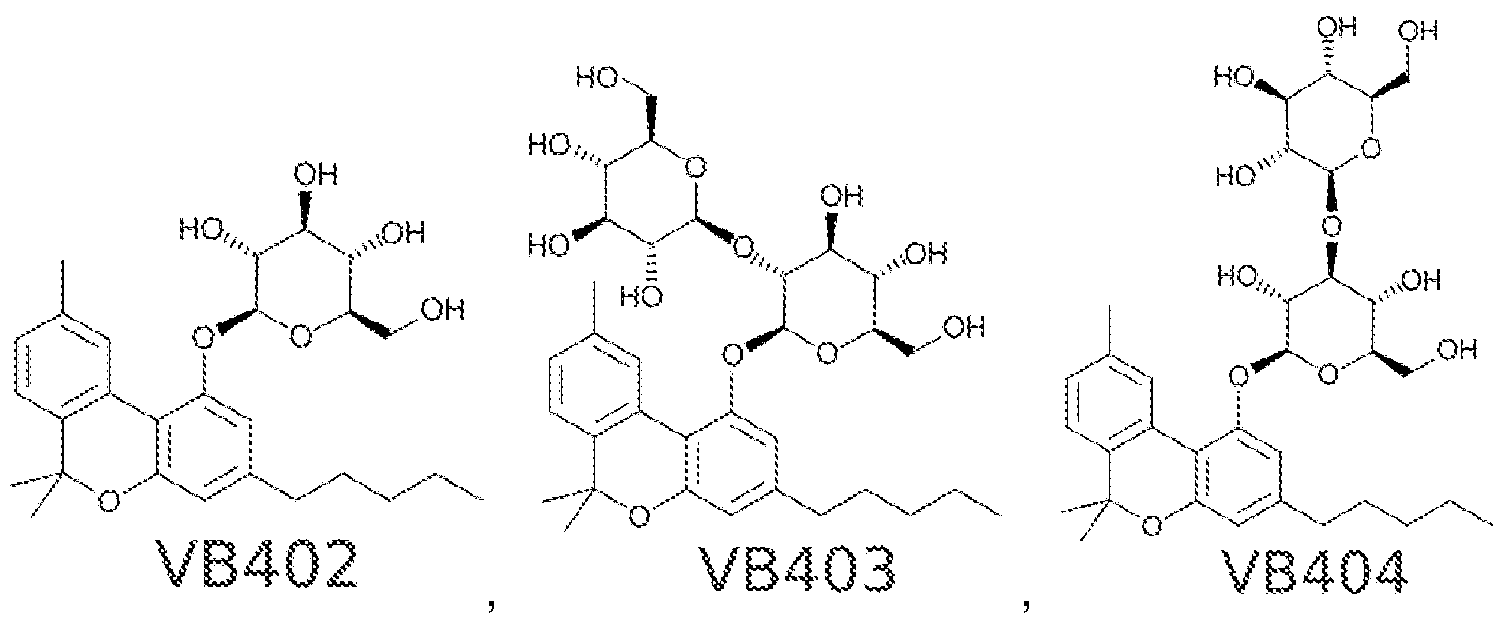
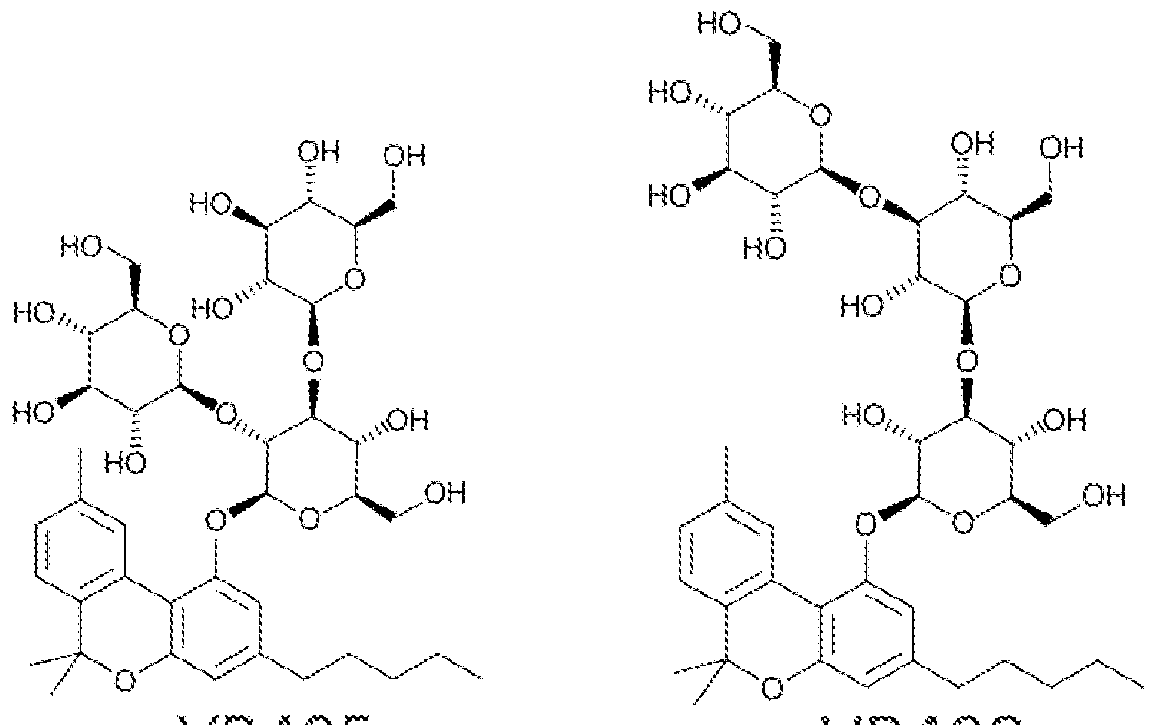
Sweet!https://patents.google.com/patent/WO2017053574A1/en "cannabinosides", glycosylated cannabinoids
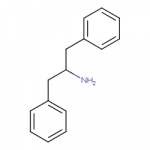
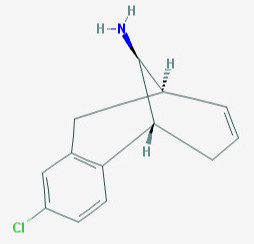
Reminds me of Org 6582:

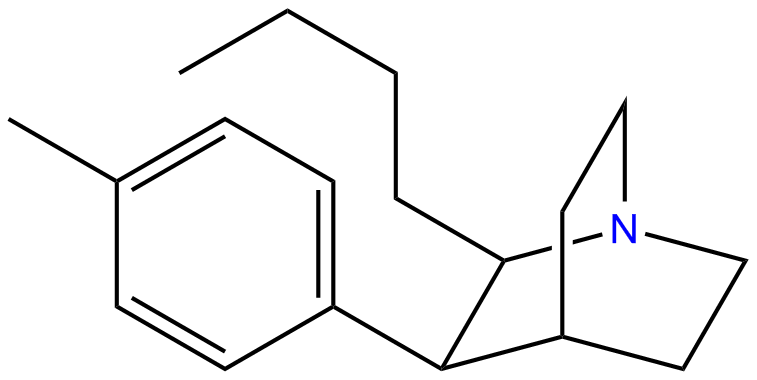
or Butyltolylquinuclidine:

Those have a quite different connectivity, because the shortest distance between phenyl and nitrogen is only two carbon atoms. The compound in my post is more like 4-benzylpiperidine.