blueberries
Bluelighter
14's a pretty magic spot for opioids. It sems like you just stick anything on there and it's a winner. Case in point; 14-PPOM.
N&PD Moderators: Skorpio
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
I Like to Draw Pictures of Random Molecules
blueberries
Bluelighter
14's a pretty magic spot for opioids. It sems like you just stick anything on there and it's a winner. Case in point; 14-PPOM.
Nagelfar
Bluelight Crew
That sweet spot oxygen on those morphinans, has it ever been just given a nitrogen? or a sulfur with crazy direct attachments.
My thought as morphine family goes is 6-(every hard pressed ether) 3-(every rapidly cleaved ester) in a cocktail with the top twelve or so HCl or perhaps even protonated differingly a la Adderall's 4 salts. Somehow I think that would be warmer, quaint, and more natural (well, spans it wider) than some other semi-synthetics. 3-(easily cleaved and more solute) 6-(hard to cleave without blocking coupling) morphine. Call it maybe multilaudiphine after laudinum
Deleted member 170540
Bluelight Crew
That sweet spot oxygen on those morphinans, has it ever been just given a nitrogen? or a sulfur with crazy direct attachments.
There are compounds like beta-naltrexamine, with an amino nitrogen on 6-carbon, and nalfurafine with amide nitrogen there, but they are usually not agonists at mu receptor (they're either mu blockers, kappa agonists or both).

Functionalization of the Carbonyl Group in Position 6 of Morphinan-6-ones. Development of Novel 6-Amino and 6-Guanidino Substituted 14-Alkoxymorphinans
The well-known opioid agonists, oxycodone and oxymorphone, and the opioid antagonists, naloxone and naltrexone, are commonly used clinical agents and research tools in the opioid field. They belong to the class of morphinan-6-ones, and produce their pharmacological effects by interacting with...
www.eurekaselect.com
Then there's the strange compound N-naphthoyl-β-naltrexamine, which targets mu-kappa receptor dimers without having much of the side effects of either mu or kappa agonists (but seems to produce some place aversion at high doses).
And if you meant a 14-amino substituent, then there are these 14-cinnamoylamino compounds, some of which are buprenorphine-like partial agonists: https://link.springer.com/chapter/10.1007/128_2010_89
Maybe there's some easier way to get a 14-hydroxy on codeine than full conversion to oxycodone... Then it would be easy to make those phenylalkanoate esters Sekio posted about.
Last edited:
Nagelfar
Bluelight Crew
Yes I meant 14, the 6 position was my addition since I can't find a chemical drawing prog. for my phone
Deleted member 170540
Bluelight Crew
Codeinone can be air oxidized to 14-hydroxycodeinone with a heavy metal catalyst, but I'm not sure if this can also be done to codeine or morphine. The cinnamic acid ester of oxycodeinone is about the potency of fentanyl: https://en.wikipedia.org/wiki/14-Cinnamoyloxycodeinone
Nagelfar
Bluelight Crew
Do that cinnimate 14 to acetorphne see what happens, that, or nearest in synth., is my imaginary pic contribution: edit the pesky endoetheno bridge by inverting the ring one space removed, (up one side, down other) but who knows what that'd do. New biomimetic synth of morphine anyone?
Last edited:
sekio
Bluelight Crew
The cinnamic acid ester of oxycodeinone
I suspect that it's actually oxycodone cinnamate, i.e. the double bond is reduced.... the same paper that details the other 14-esters has 14-cinnamate as 175 +/- 75 x morphine potency (oxycodone itself at 0.3x).
I was under the impression that codeinone and 14-hydroxycodeinone are crap analgesics.
Last edited:
Nagelfar
Bluelight Crew
Do that cinnimate 14 to acetorphne see what happens, that, or nearest in synth., is my imaginary pic contribution: edit the pesky endoetheno bridge by inverting the ring one space removed, (up one side, down other) but who knows what that'd do. New biomimetic synth of morphine anyone?
Came to the library (shift + PrtScn-SysRq & ctrl v have been the same since the '90s, yay for me. Getting old)
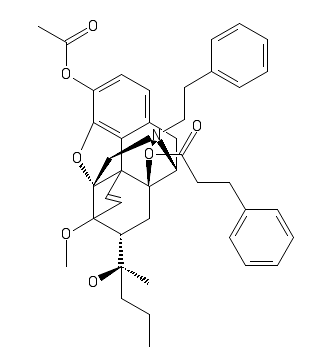
(Edit: change above by putting the bottom bridge end to the seven position where the alcohol is, so it's symmetrical)
And because I can't resist the speedball:
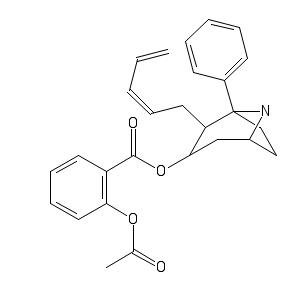 Last edited:
Last edited:
Deleted member 170540
Bluelight Crew
I suspect that it's actually oxycodone cinnamate, i.e. the double bond is reduced.... the same paper that details the other 14-esters has 14-cinnamate as 175 +/- 75 x morphine potency (oxycodone itself at 0.3x).
I was under the impression that codeinone and 14-hydroxycodeinone are crap analgesics.
You can even make that cinnamoyl ester from naltrexone and it will be a weak mu partial agonist. The 3-methylated codeine-like version of that has more of agonist effect:
Nagelfar
Bluelight Crew
I just did a search and found a 1992 findings article on pethidine derivatives made with a tropane substitution, I can't figure how to copy words and addresses with my phone so if someone has access to the article (top of Google search nearly for me) would someone so willing post one or two here for me?
The words "pethidine", "opioid" and "tropane" seem to work
Also: if you go to molview.org and type in Anthraxiton you get a strange compound. WP has no such entry. Could somebody identify it?
sekio
Bluelight Crew
Anthraxiton is brand name for sodium salt of diclofenac which is shown
I believe this is the article you are referring to.
I just did a search and found a 1992 findings article on pethidine derivatives made with a tropane substitution, I can't figure how to copy words and addresses with my phone so if someone has access to the article (top of Google search nearly for me) would someone so willing post one or two here for me?
The words "pethidine", "opioid" and "tropane" seem to work
Nagelfar
Bluelight Crew
Thank you sekio
Anthraxiton is brand name for sodium salt of diclofenac which is shown
Last edited by a moderator:
Nagelfar
Bluelight Crew
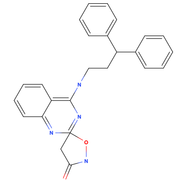
Above the synthesis of both those allosteric modulators at DAT in my recent threads nobody has responded to yet. ; -P
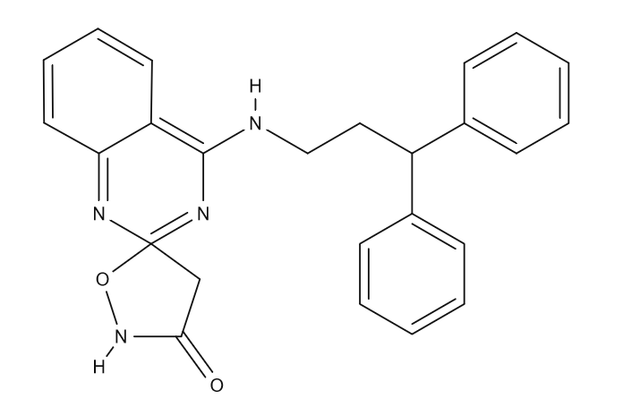
compound 11a (PMID: 23022052 Click here)
+
compound SoRI-20041 (PMID: 19244097 Click here)
I made sure to overlay with how the tropanyl in the one would match the benztropanes analogy in the other. Viable as *anything* that can be said for QSAR having theoretical probability (which I believe is null in most cases, but certainly not in divergent drugs spliced into one portmanteau)
Last edited:
sekio
Bluelight Crew
That has a Texas carbon. Not sure how stable a hydroxy/amidine (apparently a carbamimidic acid? thanks chemdraw) would be either?
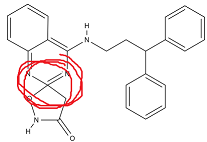 Last edited:
Last edited:
Nagelfar
Bluelight Crew
The middle arene beside the penta-spiro and opposite other aryl? That what it's called? Yeah just now wishing I could rotate the faux pi-symmetry cake walk / Chinese firedrill style one carbon around.
That has a Texas carbon.
Oh yes the fifth link on the spiro, I just thought it'd look cleaner but yeah it's still be five. At least Texas, as you say, is between the hydroxy and the nitrogens then ; -j : -P
Last edited:
sekio
Bluelight Crew
I would think that the compound with the "right" structure would rearrange to a N-hydroxy-amide...
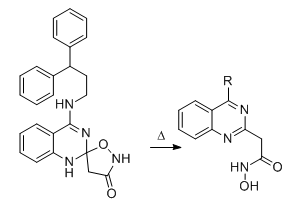
Nagelfar
Bluelight Crew
All cyclopentanes or just the above? Other ones from the paper, such as the one that works on SERT may be more viable? And if you mean my verbal correction of it I was already beginning to think I was right the first time around.
Deleted member 170540
Bluelight Crew
Just another thought about the 3-methyl-14-cinnamoyl-naltrexone: if this is almost a full mu agonist, and the 3-demethylated metabolite is almost a full antagonist, it could possibly cause some kind of 'hangover' even after single dose because the activation of mu receptors is suddenly replaced with blockage of them.
And start redosing at that point and you're soon in real trouble...
Nagelfar
Bluelight Crew
^poor naive sod who starts that regimen unknowingly.