Ondansetron is a highly specific and selective serotonin 5-HT3 receptor antagonist, with low affinity for dopamine receptors. The 5-HT3 receptors are present both peripherally on vagal nerve terminals and
centrally in the chemoreceptor trigger zone of the area postrema. Serotonin is released by the
enterochromaffin cells of the small intestine in response to chemotherapeutic agents and may stimulate
vagal afferents (via 5-HT3 receptors) to initiate the vomiting reflex. It is thought that ondansetron's antiemetic action is mediated mostly via antagonism of vagal afferents
with a minor contribution from antagonism of central receptors.
[19]
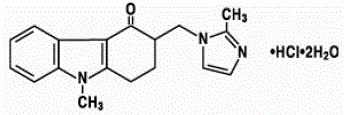
Ondansetron, marketed under the brand name Zofran, is a medication used to prevent nausea and vomiting caused by cancer chemotherapy, radiation therapy, or surgery.
[1] It is also useful in gastroenteritis.
[2][3] It has little effect on vomiting caused by motion sickness.
[4] It can be given by mouth, by injection into a muscle or into a vein.
[1]
Common side effects include diarrhea, constipation, headache, sleepiness, and itchiness. Serious side effects include
QT prolongation and severe allergic reaction. It appears to be safe during pregnancy but has not been well studied in this group. It does not have any effect on dopamine receptors or
muscarinic receptors.
[5]
Ondansetron was first used medically in 1990.
[6] It is on the World Health Organization's List of Essential Medicines, the most effective and safe medicines needed in a health system.
[7] It is available as a generic medication.
[1]
Ondansetron is well absorbed from the gastrointestinal tract and undergoes some first-pass metabolism. Mean bioavailability in healthy subjects, following administration of a single 8-mg tablet, is approximately 56%.
Ondansetron systemic exposure does not increase proportionately to dose.
AUC from a 16-mg tablet was 24% greater than predicted from an 8-mg tablet dose. This may reflect some reduction of first-pass metabolism at higher oral doses. Bioavailability is also slightly enhanced by the presence of food but unaffected by antacids.
Ondansetron is extensively metabolized in humans, with approximately 5% of a radiolabeled dose recovered as the parent compound from the urine. The primary metabolic pathway is hydroxylation on the indole ring followed by subsequent glucuronide or sulfate conjugation. Although some nonconjugated metabolites have pharmacologic activity, these are not found in plasma at concentrations likely to significantly contribute to the biological activity of ondansetron.
In vitro metabolism studies have shown that ondansetron is a substrate for human hepatic cytochrome P-450 enzymes, including CYP1A2, CYP2D6, and CYP3A4. In terms of overall ondansetron turnover, CYP3A4 played the predominant role. Because of the multiplicity of metabolic enzymes capable of metabolizing ondansetron, it is likely that inhibition or loss of one enzyme (e.g., CYP2D6 genetic deficiency) will be compensated by others and may result in little change in overall rates of ondansetron elimination. Ondansetron elimination may be affected by cytochrome P-450 inducers.
[Safe to take with MAOI??]
PRECAUTIONS: DRUG INTERACTIONS. <===[Keep in mind that a lot of those folks were on serious chemo meds that had bad reactions]
Serotonergic Drugs:
Serotonin syndrome (including altered mental status, autonomic instability, and neuromuscular symptoms) has been described following the concomitant use of 5-HT3 receptor antagonists and other serotonergic drugs, including selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs) (see WARNINGS).





 I read the PD forums here like its my job and I respect your informed opinions!! And I didn't see a Big&Dandy ondansetron thread might it be appropriate?
I read the PD forums here like its my job and I respect your informed opinions!! And I didn't see a Big&Dandy ondansetron thread might it be appropriate?