We're on the same team, if I come off as arrogant I apologize.
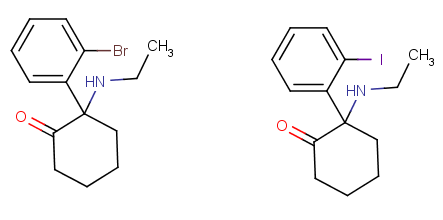
Ketamine is just right, NENK's ethyl shoves an extra carbon and two hydrogens in one location of the molecule thats already crowded, and this overcrowds it so potency goes down.
Then "2-MeO-Ketamine" takes the Ketamine molecule, which has one chlorine atom, and replaces it with a methoxy, thats 5 (!) atoms. Again overcrowding ensues. Yes you can often replace a chlorine with a methoxy and retain activity but compare DOC and TMA-2, thats the same swap job and the methoxy compound is only 1/10 as potent, give or take, this without any crowding.
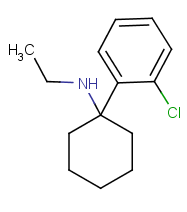
Now MXE: a 3-methoxy group is conveniently out of the way and retains potency, now you can enlarge your methyl to an ethyl and arrive at eticyclidine-like enhanced potency. If you were to migrate the methoxy back to the 2 position the molecule would be even worse than 2-MeO-Ketamine as for potency because its even more crowded.
3-MeO-PCP retains activity, 2-MeO-PCP would be crap. 4-MeO-PCP is an experientally pleasurable compound but potencywise it takes a nutshot, its about 1/10 of PCP's potency if not less so. Here it doesnt crowd the area between the rings but it sits awkwardly on the receptor, as do just about all para-phenyl arylcyclohexylamines. 4-MeO is pleasurable but for a class of drugs thats typically active in the lower milligram to higher microgram levels its very low potency.
I'm high, I hope this makes sense. I don't want to hide "my" molecule, but I want it to succeed and that takes the right person at the right time, in an otherwise clueless world.
We got to heed the advice we get from within. If I was told to tell the world I'd post it openly.
Its important to realize I didnt think up my molecule, I channeled it, as a young teen holing out on Ether, it just happens to make a whole lot of sense to the grownup me who knows a bit of chemistry.