-
Psychedelic Drugs Welcome Guest
PD's Best Threads Index Social ThreadSupport Bluelight Psychedelic Beginner's FAQ
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
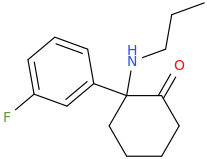
Novel dissociative 3-Fluoro-2'-Oxo-PCPr
- Thread starter roi
- Start date
Atmozfears
Bluelighter
- Joined
- Aug 14, 2015
- Messages
- 207
My god, those names. Any new dissociatives is good news, though! 

All I see is "PCP"........ lol. Interesting, I know quite a few will be watching this.
That's PCPr... Pr being short for Propyl, so it's a chain, not a ring like in PCP.
In other words they were going for structural similarities to Ketamine and MXE.
Incunabula
Bluelighter
- Joined
- Dec 10, 2010
- Messages
- 1,862
That's PCPr... Pr being short for Propyl, so it's a chain, not a ring like in PCP.
In other words they were going for structural similarities to Ketamine and MXE.
No, not ketamine. The halogen is in 3 position on the phenyl ring, and not on the 2 position like in ketamine. Considering NENK there's a strong indication that the latter would be inactive. The only structural similarity of this one to ketamine is the 2-oxo, so this one will probably be more a MXE or O-PCE type dissociative - if it's any good at all. I think PCPr was supposed to be more sedative than PCP and PCE btw.
I wonder why they didn't make the 3-MeO instead of 3-fluoro? imo that would have been more promising.
roi
Bluelighter
- Joined
- Sep 2, 2013
- Messages
- 1,545
It's the first PCPr analogue in possibly decades, however I doubt that N-propyl will be very different compared to N-ethyl in both potency and effects, should be quite close.
The big unknown is the electronegative fluorine at the meta position. We know that halogens at the ortho position (ketamine, fluorodeschloroketamine) greatly reduces potency. But so does a ortho-methoxy group as well (methoxyketamine), while that doesn't appear to be the case at the meta position at all (3-MeO-PCE and 3-MeO-PCP are not far from PCE/PCP potency).
The big unknown is the electronegative fluorine at the meta position. We know that halogens at the ortho position (ketamine, fluorodeschloroketamine) greatly reduces potency. But so does a ortho-methoxy group as well (methoxyketamine), while that doesn't appear to be the case at the meta position at all (3-MeO-PCE and 3-MeO-PCP are not far from PCE/PCP potency).
N0 W4RN1NG
Bluelighter
- Joined
- Jul 16, 2008
- Messages
- 506
2-FK is rumored to be more effective than the ortho-methoxy or even ortho-chloro counterparts, despite them all sharing relatively similar electronegativity.
So OP is correct to assume we will be dealing with something similar to the 3-MeO homologues, and perhaps at an even more appreciable level.
I disagree that the lengthening of the alkyl chain will be negligible though. I think this this will require amounts closer to 2-oxo(2-chloro-PCM) than to 2-oxo(3-MeO-PCE).
I'm still waiting for the PCiP derivs to arrive. Those are projected to be superior to any other N-substitution, if only by bonding affinity - I have no knowledge of efficacy there.
Also, Roi, it's so saddening to see someone like you, with great potential, to be so adamantly devoted to the indirect downfall of our little world. I know it's a bit hypocritical of me to say the following, but if you choose to respond to this portion of my reply, please do so via PM. I'm only doing this in the thread because I'm on a mobile device and my PM functions aren't working correctly.
So OP is correct to assume we will be dealing with something similar to the 3-MeO homologues, and perhaps at an even more appreciable level.
I disagree that the lengthening of the alkyl chain will be negligible though. I think this this will require amounts closer to 2-oxo(2-chloro-PCM) than to 2-oxo(3-MeO-PCE).
I'm still waiting for the PCiP derivs to arrive. Those are projected to be superior to any other N-substitution, if only by bonding affinity - I have no knowledge of efficacy there.
Also, Roi, it's so saddening to see someone like you, with great potential, to be so adamantly devoted to the indirect downfall of our little world. I know it's a bit hypocritical of me to say the following, but if you choose to respond to this portion of my reply, please do so via PM. I'm only doing this in the thread because I'm on a mobile device and my PM functions aren't working correctly.
Hexagon Sun
Bluelighter
- Joined
- Mar 29, 2010
- Messages
- 737
Is this one available?. Any speculations in potency and duration?
3-fluoro-2-oxo-pce could be a decent MXE analog too
3-fluoro-2-oxo-pce could be a decent MXE analog too
Synthetic_Night
Greenlighter
- Joined
- Jan 16, 2016
- Messages
- 33
I will get some soon, i will report when i have tested it
Help?!?!
Bluelighter
- Joined
- Oct 7, 2009
- Messages
- 5,071
No that's PCPy unfortunately! It was said to have barbiturate like sedation. Sounds very nice but r was stimulating.No, not ketamine. The halogen is in 3 position on the phenyl ring, and not on the 2 position like in ketamine. Considering NENK there's a strong indication that the latter would be inactive. The only structural similarity of this one to ketamine is the 2-oxo, so this one will probably be more a MXE or O-PCE type dissociative - if it's any good at all. I think PCPr was supposed to be more sedative than PCP and PCE.
Incunabula
Bluelighter
- Joined
- Dec 10, 2010
- Messages
- 1,862
No that's PCPy unfortunately! It was said to have barbiturate like sedation. Sounds very nice but r was stimulating.
Yes, know I remember, you're right of cause
I will get some soon, i will report when i have tested it
Should be interesting!!!!
Synthetic_Night
Greenlighter
- Joined
- Jan 16, 2016
- Messages
- 33
My substance has arrived, i will shortcut it with floxy. What do you think is a good starter dose? Does someone knows for 2-oxo-pcpr? Should nearly be in that range, probably a bit lower in potency...ergo a little bit higher dosage
Solipsis
Bluelight Crew
- Joined
- Mar 12, 2007
- Messages
- 15,509
Why don't you titrate from really really low without redosing that same day (tolerance would screw it up), always a smart idea but here it's different enough from other known-well-enough compounds to just be safer rather than sorry. Being among the first to try it carries with it some risks and conditions, including not being impatient. I get it if you don't start at 1 µg but trying to start at the dose range you guesstimate it being active at is really stupid.
Actually 3-MeO-PCPr was commercially available for a brief period of time alongside 3-MeO-PCPy and the first batch of 3-MeO-PCE from the UK. This was in 2011, I think.. 3-MeO-PCPr was indeed almost indistinguishable from it's N-Ethyl counterpart in it's qualitative effects as well as the dosage needed.. If memory serves me right 25mg lead to a smooth +++.. Anyways I am excited how 3-Fluoro-2'-Oxo-PCPr will turn out...It's the first PCPr analogue in possibly decades, however I doubt that N-propyl will be very different compared to N-ethyl in both potency and effects, should be quite close.
Synthetic_Night
Greenlighter
- Joined
- Jan 16, 2016
- Messages
- 33
so 3-meo-pcpr was around 25mg.... ok, it seems the alkyl-substitute connected to the amine leads to different potency and duration- methoxMetamine needed twice the dosage of MXE. I was thinking, an propyl substituent is going the other way and even enforces the potency. On the phenyl-ring is in position 3 a fluor. This is clearly different from methoxy-groups, fluor is more like an ordinary bound hydrogen with stronger resistance for metabolic oxidation. And even the oxo-group plays an important role i think, it enhances serotonergic effects, while no oxo on the cyclohexane and a methoxy on the phenyl-ring is more responsible for an opioid effect. Removing the halogen (chlor) from ketamine leads to more lucid effects, and even an significant increase in potency. This can be seen on O-PCM. I think with a fluor floxy will be overall between Ketamine and O-PCE within its effects and dosage. Well, this are my thoughts about structure-effect-relationships, i will know more in around an hour :D I will keep you updated
€: i insufflated 5mg...it burns a lot, holy shit :D hydrochlorides are well known for that..... it reminds me of good old 2C-E nose pain oO next time oral is the way to go
€: i insufflated 5mg...it burns a lot, holy shit :D hydrochlorides are well known for that..... it reminds me of good old 2C-E nose pain oO next time oral is the way to go
MSK
Bluelighter
- Joined
- Apr 15, 2015
- Messages
- 651
so 3-meo-pcpr was around 25mg.... ok, it seems the alkyl-substitute connected to the amine leads to different potency and duration- methoxMetamine needed twice the dosage of MXE. I was thinking, an propyl substituent is going the other way and even enforces the potency. On the phenyl-ring is in position 3 a fluor. This is clearly different from methoxy-groups, fluor is more like an ordinary bound hydrogen with stronger resistance for metabolic oxidation. And even the oxo-group plays an important role i think, it enhances serotonergic effects, while no oxo on the cyclohexane and a methoxy on the phenyl-ring is more responsible for an opioid effect. Removing the halogen (chlor) from ketamine leads to more lucid effects, and even an significant increase in potency. This can be seen on O-PCM. I think with a fluor floxy will be overall between Ketamine and O-PCE within its effects and dosage. Well, this are my thoughts about structure-effect-relationships, i will know more in around an hour :D I will keep you updated
€: i insufflated 5mg...it burns a lot, holy shit :D hydrochlorides are well known for that..... it reminds me of good old 2C-E nose pain oO next time oral is the way to go
Interesting! Looking forward hearing back from you again! Have you used other dissos like 3-MeO-PCP or 2-OxO-PCE in order to compare to them? Also, could you please clear your PM inbox, as I wanted to contact you, but your inbox is full?
Synthetic_Night
Greenlighter
- Joined
- Jan 16, 2016
- Messages
- 33
Yes, my inbox is full. Only 5 messages in my folder and that's it ^^ but now it is empty.
But i think this substance is not really working for now....maybe a bit like an opioid, but i am not recognizing much....maybe i will test again in 2 days, nothing more will happen today
But i think this substance is not really working for now....maybe a bit like an opioid, but i am not recognizing much....maybe i will test again in 2 days, nothing more will happen today