cegli
Bluelighter
- Joined
- Apr 14, 2008
- Messages
- 331
Does anyone have any thoughts on this chemical?
2-ethyl-3-phenyl-4(3H)-quinazolinone (Methaqualone Analog)
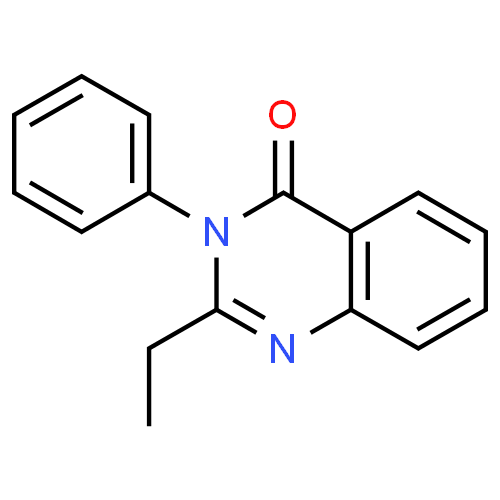
It's being called Ephinazone for reasons unknown to me. It's basically Methaqualone, with the 2-methyl on the quinazolinone ring extended to a 2-ethyl, and the methyl on the phenyl ring removed.
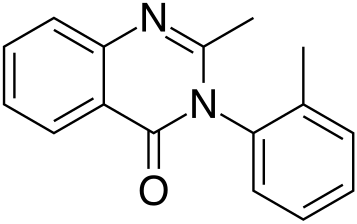
Methaqualone for comparison:
I'm trying to figure out the logic of this one, and all I have is some (probably inaccurate) musings. So far from a SAR point of view we've seen:
Changes to 2-methyl:
Ethyl - Etaqualone - Too short, effects not as good as Methaqualone.
Bromine - Mebroqualone - Too short, effects not as good as Methaqualone.
Chloro -Mecloqualone - Used as prescription drug, never took off like Methaqualone. Short and there's probably a good reason why it was never too popular.
Other:
4-methylmethaqualone - Heard it was somewhat decent, but can cause seizures.
I've never seen a single analogue with nothing on the phenyl ring. Lengthening the 2-methyl to a 2-ethyl has been done before in Chloroqualone, but with the di-chloro on there, you can't really get a good idea of the SAR.
Now here's where I'm really out of my league and may make a fool of myself. Why would you remove the methyl from the phenyl and tack it onto the quinazolinone? Why wouldn't you first try just lengthening the methyl on the quinazolinone first, or just remove the methyl from the phenyl? Take each change one step at a time, to observe the SAR and not get too far from Methaqulone.
My only guess was to keep the bulk the same and the electronegativity similar for that region of the molecule?
Remember I have no chemistry background, so go easy on me .
.
Edit: Just noticed that Ethinazone was a brand name for Etaqualone, so Ephinazone is some kind of play on that.
2-ethyl-3-phenyl-4(3H)-quinazolinone (Methaqualone Analog)
It's being called Ephinazone for reasons unknown to me. It's basically Methaqualone, with the 2-methyl on the quinazolinone ring extended to a 2-ethyl, and the methyl on the phenyl ring removed.
Methaqualone for comparison:

I'm trying to figure out the logic of this one, and all I have is some (probably inaccurate) musings. So far from a SAR point of view we've seen:
Changes to 2-methyl:
Ethyl - Etaqualone - Too short, effects not as good as Methaqualone.
Bromine - Mebroqualone - Too short, effects not as good as Methaqualone.
Chloro -Mecloqualone - Used as prescription drug, never took off like Methaqualone. Short and there's probably a good reason why it was never too popular.
Other:
4-methylmethaqualone - Heard it was somewhat decent, but can cause seizures.
I've never seen a single analogue with nothing on the phenyl ring. Lengthening the 2-methyl to a 2-ethyl has been done before in Chloroqualone, but with the di-chloro on there, you can't really get a good idea of the SAR.
Now here's where I'm really out of my league and may make a fool of myself. Why would you remove the methyl from the phenyl and tack it onto the quinazolinone? Why wouldn't you first try just lengthening the methyl on the quinazolinone first, or just remove the methyl from the phenyl? Take each change one step at a time, to observe the SAR and not get too far from Methaqulone.
My only guess was to keep the bulk the same and the electronegativity similar for that region of the molecule?
Remember I have no chemistry background, so go easy on me
Edit: Just noticed that Ethinazone was a brand name for Etaqualone, so Ephinazone is some kind of play on that.
Last edited: