Mm. Okay. Well I will try to keep this simple then.
If a drug is contaminated, then the most likely culprit will be a second chemical, piggybacking on the delivery method. In the case of acid this delivery method is assumed to be blotter, since that's often the context in which dirty acid is referred to. So naturally one wishes to know: what other compound, and how much of it?
Taking 100 micrograms as the average dose of LSD on a blotter, we can see that the amount of contaminant would have to be small – incredibly small. And yet paradoxically it must be enough to create negative symptoms in body, mind, or both. So let's say the acid is 80% pure. This would be highly unlikely I suspect, as you don't get around to making acid with any less than truly first-class chemistry skills. But for the sake of argument, 80% impure it is.
That means 80 mics of acid, 20 of 'other stuff'. So our hypothetical contaminant, the dirt in 'dirty acid', would need to exert its action upon the human animal at a dose of *20 micrograms*! That's not even a threshold dose were we speaking about LSD itself, and LSD is one of the most potent psychedelic chemicals known.
But it goes further. And this gets to itsALLfake's question earlier: what we are feeling when we take acid is actually the effect of one of four isomers: (+)-LSD is the only active isomer, (-)-LSD and (+)-iso-LSD and (-)-iso-LSD are all inactive compounds. Now I do not know what the ratios are, it is likely that at least some of the acid that is around is racemic. This means all of the isomers, in equal proportions. Or possibly there may be stereoselective syntheses that can push those ratios towards the active isomer to one extent or another.
If the blotter is loaded with racemate, then it would follow be that there is actually 400 mics of LSD isomers, one quarter of which would be the (+)-LSD. In that case 80% pure would mean that there could be actually 320 mics of racemic LSD, one quarter of which is the active isomer, and 80 mics of contaminant. This is slightly more reasonable as far as finding chemical candidates that are this potent.
I make this point in spite of my ironclad surety that dirty acid is a myth. I think the sum total of the evidence backs up my side of the argument, and I haven't heard anybody on the other side of the debate so much as suggest a mechanism for dirty acid. The racemate-loaded blotter as means for explaining why a contaminant wouldn't have to be THAT potent is meant as proof of looking objectively at the issue. If I'm willing to go farther than anybody on *the other side of the debate* by actually positing such caveats that make their arguments less ridiculously unlikely, then hopefully people wouldn't ascribe me to be unfairly judging evidence, right? Or on the other hand, if I'm not a believer and yet I'm the only one theorizing on their side of the debate, perhaps the evidence and their arguments are simply weak when looked at closely? Mmm, could go either way, but better to try to demonstrate a lack of bias despite my strong opinions on the subject than to just blunder about looking sneaky and bent.
Anyway, getting on with the explanation: there is a second criteria for the contaminant, should it exist. It would need to be produced by a sloppy acid synthesis. This is a major sticking point, every bit as much as the wildly unlikely potency, for several reasons. First, as I states, amateurs don't synthesize acid. Second, the people that CAN synthesize acid will know when things go wrong. Third, even if they didn't, you don't fuck up your synth and get 'almost acid but not really acid that is still active and makes you feel shitty'. You get a bunch of garbage, and your chromatography column will show this garbage to be lacking in acid, and the jig will be up. And fourth, we know lysergamide chemistry, speaking from the standpoint of the sum knowledge of mankind. There are not 'other lysergamides' that come out of botched or low-yield acid syntheses.
So reviewing the conditions, we have something that must be exquisitely potent, more so than acid itself, must produce symptoms in the human animal, must be made as a byproduct of highly unlikely sloppy, low-yield syntheses, and must Be invisible to analysis by blotter-testing concerns. There is not a single compound I know of that is a possible fit for these characteristics. And nobody with a better handle on lysergamide chemistry than me is willing to make suggestions since they consider the matter to be such a nonissue as to be not worth their time (see Ebola?'s response on page one). The people who do believe in dirty acid fail to come up with candidates either, or theorize mechanisms for 'dirtiness' that wouldn't require invoking highly unlikely compounds.
On the other hand, acid is known to be just about the most variable psychedelic drug we have. It is also known to be occasionally hard on the body, in the manner of its less pleasant (read: dangerous / possibly deadly) cousins, such as vasoconstriction. Third acid is incredibly powerful, and has a propensity for what is known as 'psychosomatic effects', where mental problems are sort of transferred to the form of general bodily malaise.
When all this is examined, I think the answer is clear, but many disagree. Decide for yourself, I guess.
---
Also: itsALLfake
On isomers: I failed to dig up anything useful to actually, you know, substantiate the lack of activity of the other isomers of LSD. However, I can explain to you in general terms why isomers differ in activity, and then generalize this to acid.
It happens in organic chemistry that you'll have alkanes, saturated carbon chains. Each atom has four substituents, counting its links to the other carbons in the chain. Carbon forms tetrahedral bond patterns when there is no strain, ring strain or whatever, on the bonds. So if you have a carbon chain, and we pick one in the middle, then there are two other atoms connected to it.
If those other two substituents are different, then you get a chiral center. To really explain this right, we need pictures:
First, you need to know what the hashed bond and the black wedge mean. I said carbon would have it's substituents arranged in a tetrahedral arrangement assuming it had no double bonds. So that means that if we look at a carbon like that above, and two bonds are perpendicular to our view – that is, we are seeing the bonds drawn as plain lines from an angle 90 degrees, so that they lie along the two-dimensional surface that is our computer screen – necessarily the other two bonds can not lie in that same plane. If they did this would not be a representation of a 3-dimensional tetrahedron.
So of those two bonds that do not lie within the plane on our computer screen, one points away from the carbon, and away from the viewer, this is the hatched bond. The other must stick out towards the viewer, and that is the black wedge.
If you're having issues understanding this, try this: take a piece of paper. Draw an equilateral triangle on it, with equally sized equilateral triangles attached to each of the three sides. Cut out along the outsides of this whole mess of four triangles. Now, fold up the triangles on the edges so that you have a pyramid. This pyramid should have four equal faces each shaped like an equilateral triangle, and four points, right? Now hold this pyramid so that two of the points are equally far away from you, in a line. Notice where the other two points now are forced to be? One is close to you, one further away. These points represent the atoms connected to the carbon, which would be located in the center of the pyramid.
So assuming you either get it the first time or (no shame in this) prefer to cut out the shapes to make the pyramid and see it for yourself, I'll continue as if this issue of what the wedges and hashes represent is understood.
Notice in the picture above, there are two molecules. There's also a dotted line down the middle, between them. This is an axis of reflection: a mirror. If you look closely, because the substituents on the wedge and hashed bond are not the same, the two molecules above, while structurally containing the same atoms, cannot be made into one another by turning them 180 degrees. Because if you turn it 180 degrees, what was the bond facing away from you is now the bond facing towards you. Whereas both molecules pictured have the same substituents on the far-away side.
The only way you could make these molecules identical is by mirroring one of them, which flips its symmetry. This does not happen in nature. Or under human power for that matter. So like your right and left hand, you have your fingers – analogous to the bonds that create the chiral center – in the same order. But the direction that the order progresses is opposite for each hand, and you cannot make your left hand into a right hand without taking a picture and mirror flipping it. Thus says the man who has all to often put on the wrong glove for a given hand in the winter.
Molecules like those above, structurally composed of the same stuff but in a different order, are known as enantiomers, after the ancient Greek for mirrored parts.
So what does the fact that you can have two enantiomers mean for drug biochemistry in the brain, let alone acid? It turns out that drugs, say, like amphetamines, have chiral centers, also referred to as stereocenters. And it also so happens that it matters to the brain's receptors which enantiomer is present.
The 'molecular docking maneuver' – the process by which a drug molecule binds to a receptor – is a sensitive thing. The molecule of drug is twisting and turning, not just the molecule itself spinning around in all directions and moving about erratically in Brownian motion, but also the bits of the molecule that are not rigid – in this case the aliphatic chain containing the amine that sticks out from the hexagonal aromatic ring in an amphetamine molecule – spinning around. Each of the single bonds in a molecule that isn't held in place by aromaticity will rotate around the axis of the bond, so that each atom in the aliphatic chain is spinning around and bringing its substituents with it.
The bonding between the receptor and the molecule isn't strong. It is based on hydrogen bonding, van der Walls forces, electrostatic give and take, steric bulk, all very weak attractive and repulsive forces. So the drug is spinning around and moving in a random fashion, while also contorting itself as any single bonds not stabilized by aromatic resonance rotates around its bond's axis, and this spinning twisting thing must dock with the receptor through the gentlest of attractive and repulsive forces. Needless to say, there are a lot of 'misfires', where it bangs into the active binding site but is conformationally the wrong shape to bind. And when it does bind, it matters where the substituents are located relative to the receptor.
Since there are two enantiomers of amphetamine, one with a alpha-position where there is a hydrogen 'pointing towards the viewer' and the alpha-methyl is on the side of the carbon opposite the viewer, called the levorotary isomer. This would be depicted with the alpha methyl as a hashed wedge if the aliphatic chain points to the right of the aromatic ring, like this:
The other would be the dextrorotsry form, which would have the hydrogen on the alpha carbon pointing away from the viewer and the alpha-methyl moiety pointing towards the viewer, depicted as a thick black wedge, like this:
Dextro and levorotary refer to how each of the isomers reacts to circularly polarized light of one direction or another, just for reference purposes, I won't get into that here.
Anyway, if the levorotary isomer binds to the receptor, the location of the alpha-methyl relative to the hydrogen on the alpha carbon results in a different reaction than if the dextrorotary form binds, with the alpha-methyl on the other side relative to the hydrogen. And so you get two different effects depending on which isomer is administered, with the dextrorotary form causing much of the 'pleasant' stimulant effects and the levorotary form being the culprit for much of the nastiness you get from stimulants. And as you might imagine, taking the 50/50 racemic mix results in effects from both isomers, leading to a state with some of the positives but also some of the negatives, but with neither type of effect predominating.
---
So how does this relate to acid? Well, until now we've been talking about amphetamine, a relatively small, uncomplicated drug, where both isomers are active. LSD is a big, complicated, unwieldy molecule, and it has two stereocenters, so thus four isomers to amphetamine's two isomers. And furthermore, one stereocenter affects the positioning of the whole diethylamide chain, and the other affects the positioning of an entire aromatic ring, AND the diethylamide chain attached to that ring.
We've so far examined isomerism from the point of view of an aliphatic chain, but chiral centers are not limited to this type of structure, and when there are ring systems that are not fully aromatic, possessing only a single double bond in the case of the ring that holds the C-5 stereocenter, chirality is a result of there being two substituents on a given carbon in the ring (assuming that carbon has no double bonds. One of those substituents will be 'behind' the ring if the ring is in the plane of the computer screen, and the other 'in font' of the ring. This is the same concept I was explaining earlier and we do use the hashed or fully-blacked out wedges to indicate positions, hashed where the bond points away from the viewer starting from the ring, with a black wedge indicating a bond where the substituents lies closer to the viewer than the plane in which the ring sits. I'm not going to try and explain this more in words, the following picture will hopefully suffice as this post is now long by even my unusual standards and I wish to get on to why the inactive isomers of acid are in fact inactive instead of just shitty, like the levorotary form of amphetamine.
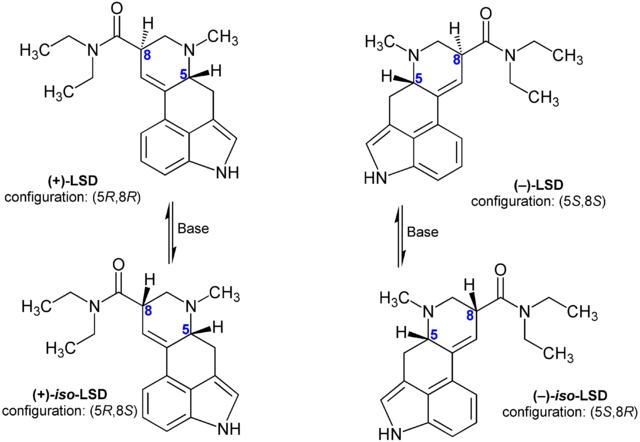
Because acid is so big, its fit in the receptor is very delicate, even more so than for amphetamine, because its got that much more shit to flop and spin around, like the diethylamide bits. Further, the two chiral centers are integral to the multi-ringed structure in one case and to the base of the diethylamide chain for the other, and the effects of the placement of the chiral centers in these locations are thus more severe than the relatively small change in the location of two terminal substituents (substituents that lack substituents themselves, a methyl and a hydrogen) on the amphetamine's aliphatic chain.
So since the molecule is so big and it's conformational situation has to be just so, and these changes alter the position of much more of the molecule than the simple switch of a hydrogen and a methyl on an amphetamine molecule, the three isomers that are not (+)-LSD are simply too much the wrong shape to bring the diethylamide portion into the position that it needs to be for binding to take place and thus for activity to be realized. This is what happens as molecules get bigger: the number of complicating factors regarding binding and conformational changes gets bigger in a likely nonlinear fashion. This is not just the result of the potential for chirality to increase or for more chiral centers to exist. That is certainly part of it, but it also has to do with the aforementioned property of single-bonded carbons that are not stabilized by aromatic resonance to twist and turn on their axis, bringing everything attached to them with it. The more complicated a molecule in other words, and the floppier and twistier it can get.
Anyway, I hope this answers your question itsALLfake, let me know if you (or anybody else) want clarification on something or other, I'd be happy to try and explain it better.







