I've done both derivative drugs at least a dozen times now, and I've taken 2C-B ~80+ times, both the HCL and HBr. I've eaten it, insufflated it, and even freebase + vaporized it; oral ingestion is by far the best RoA for 2C-B in my opinion – snorting it hurts like fucking crazy, even the hydrobromide salt… and vaporizing it induces
way more anxiety in me than simply orally consuming it.
βOH-2C-B is okay; I mostly agree with
@Buzz Lightbeer that it's kinda lackluster compared to other compounds, but it's
not without its own charms. It's a creeper; comes on more slowly than 2C-B or 2C-B-FLY, even. sometimes, I forget I've taken
βOH-2C-B (or
“BOHB” if you prefer,
rhymes with “robe”), and ~4h. later I'm in the middle of something wondering why my thoughts are all fucked up… oh yeah, the
beta-hydroxy-bromo-mescaline… And then that shit lasts forever, but I've never not enjoyed it. Despite all of this, it's the least impressive of the three compounds in question here, and the least potent mg for mg.
2C-B-FLY is absolutely worth researching and exploring. Its effects are also long-lasting and the come-up is slow. Compared to 2C-B, 2C-B-FLY is a lot less mellow. It carries more body-load for sure, and I find it's best to have physical activity to channel that into. Fortunately, it has a similar sexual-enhancing profile to the parent compound, 2C-B. Honestly, all of these compounds make for wonderful aphrodisiacs, though they're not as aggressive about it as, say, 5-MeO-DiPT, in my opinion.
What's interesting about 2C-B-FLY is that the two methoxy groups extending from the 2- and 5-position of 2C-B's benzene ring are fused into furan rings, so it produces a 2,5-difuran compound instead of a 2,5-dimethoxy. Physically speaking,
2C-B's dimethoxy setup is not as conformationally rigid as 2C-B-FLY's difuran ring setup. Think of it like the 2,5-dimethoxy molecules sort of flail about like a tube sock man, the brain flushes it out sooner because of this physical property. Hence the shorter duration of 2C-B compared to 2C-B-FLY, which tends to get sort of “wedged” into place with the receptor. This is also the reason 2C-B-FLY is more potent than its parent compound, 2C-B:
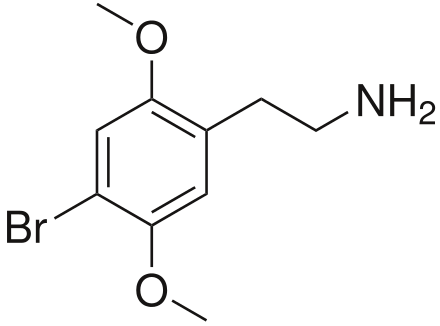
For reference,
here's 2C-B. Aka 2,5-dimethoxy-4-bromophenethylamine.
Note the flailing tube sock man methoxy groups at 2 and 5.
They are physically flexible a bit so they don't bind particularly rigidly at the receptor sites:
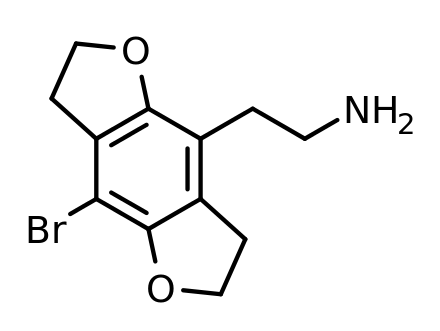
And this next one is
2C-B-FLY.
See how those oxygen molecules are held firmly in place by the ring structure?
It causes the molecule to linger at its locations of activity,
and it causes it to be more potent as well:
So whereas I might take:
60 mg of βOH-2C-B,
and I might take:
20 mg of 2C-B,
I only need about
12 mg of 2C-B-FLY.
Lotta bang for your buck there.





 exactly
exactly