Factors That Make Each MDMA Batch and Experience Unique
The 3 Main Factors:
1. Set and Setting
2. Impurities
3. Polymorphs
The 3 Minor Factors:
1. Isomers
2. Salts
3. Binders
Unfortunately we as humans are always trying to narrow down anyone one problem into a single solution. It’s just easier for us to wrap our heads around. We try to distill anything we can down to one single thing whatever that may be. But as for most things, MDMA batch and experience variability is more complicated than that. It’s not just salts, it’s not just isomers, or set/setting, it’s a combination of these above that shape what you get out of it.
Set and setting is number 1 for a reason. I believe hands down it is the biggest factor of them all no matter what people want to say. Yes in the beginning of your MDMA using career it may be that every experience is mind blowing but over time, from it’s effects to your serotonin system to nostalgia, this can become harder to achieve. This can be negated by proper diet, hydration, and supplementation when you do imbibe to keep the tolerance low for next time. As well as changing up the setting, and trying new things. (Sitting at home popping MDMA eventually becomes boring no matter who you are, and even raves can become monotonous.) I’ve had some of my best and some of my worst experiences on the same batch of MDMA, simply from a change in set and setting.
Set and setting also includes things like current diet (effecting various enzyme levels), drug intake (effecting neurotransmitters one way or another), social relationships, what side of the bed you woke up on, the list is really endless with this. One little variable that you couldn’t even imagine would effect the experience, could…
With that out of the way the rest will strictly relate to the batch of MDMA itself. Impurities are next on the list. When looking at impurities it is obvious that each synthetic route has unique impurities separate from other routes. Hence the police use this to help track synthesis trends and possibly as indicators to how much a certain producer has pushed. It has been shown in one study that certain impurities found (out of only 12 tested mind you, there’s hundreds if not thousands) were indeed relatively active when comparing to MDMA itself. (The most active ones found were from the Leuckart reaction, more about that in a second.)
Some of these impurities are most definitely active and even if they may not be as active as MDMA itself, we know from past experimentation from greats like Shulgin that certain substances can indeed potentiate even if they aren’t extremely active themselves. Some can also blunt or negate certain effects by competing with MDMA at certain receptor sites. Think of buprenorphine, doesn’t really feel that great even at high doses but will outcompete other opiates/oids for receptors. Mix 1mg of buprenorphine into 100mg of morphine and while only being about 1% of the overall mixture it will greatly effect the overall experience, same if I added a tiny bit of naloxone.
The list of active impurities is endless and depending on the chemist there could be quite a lot in there. Just a few fairly active ones off hand are; MDA, MDEA, DMMDA, N-formyl MDMA, MDDMA, PMA, PMMA. All of which can come from improper purification of certain precursors. In a perfect world MDMA would be just MDMA, but in the world of illegally synthesized drugs people take shortcuts and sometimes lots of them. If we take a look at more recent analysis of ecstasy tablets (EData) we still see pills come in fairly regularly that show large amounts of various precursors.
On another thread a user talked about how each batch of MDMA is kind of like a snowflake and got shot down for it.. He is in a way very right about that. A small change can effect the end product. Using impure homemade methylamine can cause MDA and MDDMA to be produced. Not fractional distilling the safrole oil can possibly wind you up with trace amounts of PMA/PMMA. Certain sources for nitromethane contain small amounts of nitroethane which could produce small amounts of MDEA. GC/MS is far far far from a perfect science and when analyzing MDMA they aren’t likely to catch many of the impurities which could be present.
Some of these impurities can alter the overall effect profile even at minute dosages, but there are also impurities which can increase the overall toxicity. Dihydrosafrole being one which is produced when safrole isn’t cleaned out during the synthesis, its pretty neurotoxic by the sounds of it. Another which is thought to possibly be toxic is 6-chloro-MDMA.
Now above I speak of the leuckart reaction, one which produces quite a few active impurities and was known by chemists as a “dirty route” that could at times be unpredictable. The reason it was used back then was due to ease of precursor availability, scalability (to produce large batches), and the fact that MDMA chemistry was in ways still in it’s infancy in comparison to today. It just so happens that back in the 90's this route was commonly used much more than in comparison to today. The supposed golden era of MDMA talked about by many. There is only one reference I could find to an analysis of ecstasy from that era, and they only tested one pill, that one pill was produced via the leuckart. I understand that is pretty weak evidence but when we look at the amounts of certain pills back then, they were produced by only a few large operations. People back then talk about a few main pills whereas today there’s too many to name, back then there were nowhere near as many different presses about. It’s my belief that likely if 1 pill was synthesized that way many others at that time would be too because of this. The next available analysis I could find came from China in early 2000’s and it seems while Leuckart was still in use it was becoming less used in place of other routes like reductive amination with sodium cyanoborohydride from MDP2P from piperonal via the nitrostyrene intermediate. (Of the 416 cases analyzed by GC-MS, 341 were found to likely be synthesized via reductive amination of MDP2P which was synthesized from Piperonal.) By the late 00’s the leuckart had largely become a synthetic route of the past as newer higher yielding, and better purity routes took it’s place.
This correlates well with the supposed change of MDMA people speak of from the early 90’s to the late 90’s/mid 00’s, but still leaves us wondering if this was the case what impurities would be effecting the MDMA in such a way to make a 60-75mg dose last 4 solid hours of dancing your heart out as so many users from that time claim. The evidence to show the route changed is kind of there but besides that we don’t have much else.
While we are on the 90’s MDMA topic I’d also like to say below when talking about isomers I will make an argument for that as well effecting the ecstasy experience back then. The only “evidence” to back up the claim is from a chemist I trust who took MDMA back in the 80’s/early 90’s, but the claim correlates well with the effects and dosage required back then. More on that in a bit…
The final main factor I’d like to discuss is polymorphism. Something I brought up in another thread here. Polymorphism is the ability of a drug substance to exist as two or more crystalline phases that have different arrangements and/or conformations of the molecules in the crystal lattice. It appears polymorphism and how to predict its effect on a psychoactive substance is still in its infancy regarding research. There’ve been numerous issues regarding polymorphism for pharmaceutical companies in the past, and lawsuits over it even when certain generic producers couldn’t replicate the same polymorph as a certain patent claimed.
Polymorphism can effect stability of the drug, bioavailability, and overall drug efficiency. While most simply talk about it in terms of bioavailability and efficiency I believe this can also change the overall effect profile of certain drugs, especially ones deemed recreational in some way.
Shulgin noted that there were many polymorphs (hydrates) found and even the melting points for anhydrous MDMA varied by a decent amount from researcher to researcher quoted by Shulgin. (Possibly indicating unknown polymorphs.) Each of these hydrates could affect the experience in some way, better or worse.
From one study I gathered that they produced “plate crystals” from anhydrous MDMA. These are typically hexagonal and flat. I have seen MDMA like this. Many other sources quote MDMA being “orthorhombic.” I have seen many MDMA crystals like this, typically they are very good. (Kinda look like little pieces of quartz.)
One of the best pieces of evidence I could find for possible variations due to polymorphism is this.. Strikes comments in TSII. She talks in the crystallization section about how DCM is the best for producing clean crystals even from impure freebase MDMA. Then the next paragraph goes as follows..
“The other amazing thing about DCM is that the crystals produced in it are weird. Crystals produced by ether are little and powder puffy. Nothing wrong with that per se. But the crystals produced in DCM are large, crystalline and sparkly. And there shouldn’t be difference, but SWINS swears the effects are more powerful.”
If anybody knows a thing or two about variation between batches of MDMA I’m assuming it would be strike. The above can really only mean one thing, a different polymorph has been formed when the crystallization solvent is changed.
Polymorphs are so finicky there is even reports of “disappearing polymorphs” where a lab synthesizes one polymorph and eventually it converts to another more stable form. Once this occurs small particles of the new polymorph make it into the air and can be used as nucleotides for the crystallization of further batches in that same lab. So in theory each lab could produce a different polymorph based on the previous polymorphs they’ve made and what particles they have floating in the air in their lab. (While this liking isn’t the case for MDMA it’s a good point in how easy it is for this change in crystalline structure to occur.)
I believe more research into this field will in the, hopefully near, future show us just how and why very pure MDMA can vary from one batch to the next. It seems most of the MDMA causing these problems over in Europe right now is of decently high purity so my thoughts can only go to polymorphism or possibly the “old” MDMA was impure and it was those impurities that caused a better effect compared to the highly pure MDMA available today. Somehow I doubt the latter, as there is highly pure batches out there which cause all the typical effects we seek so…
On to the minor factors..
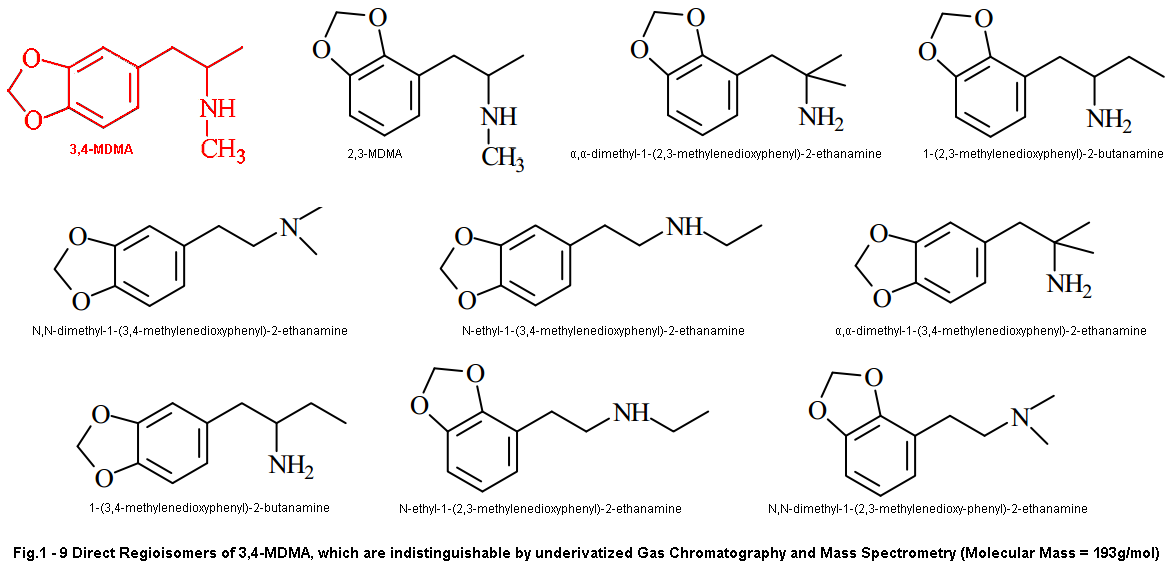
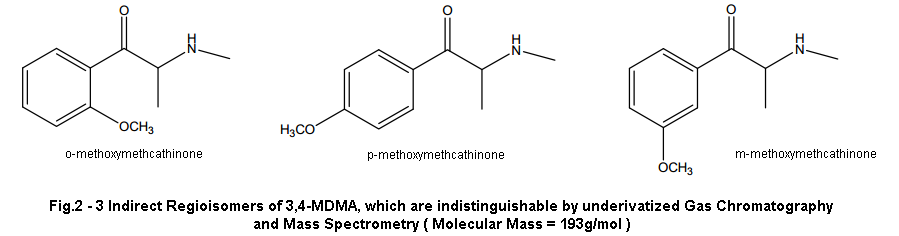
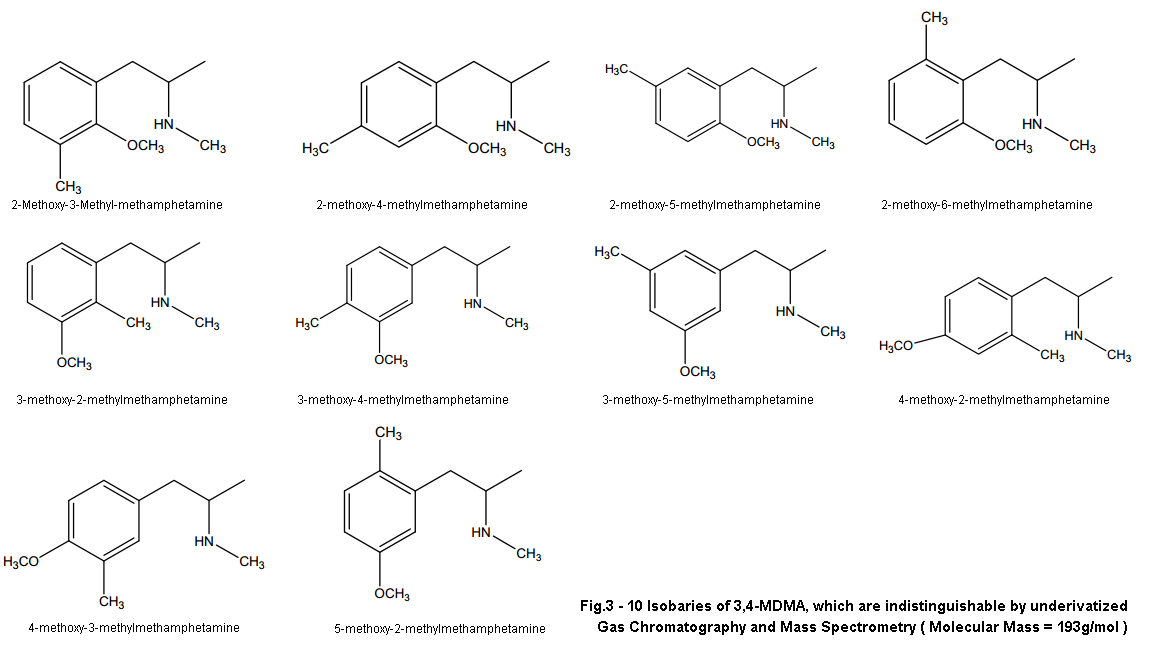
And we’ll start with the one people have been harping on for years now, even myself at certain points. That is ISOMERS! We’ve heard this same argument time and time again, and while I do believe it is possible isomers have played a role in the variation of effects over the decades I don’t see it as very likely. At least for our current “Dutch Mega Crap Pills” situation… Maybe for the reason the early 90’s pills were different but I’ll get to that in a second.
Isomers just don’t fit the profile people. I’m sorry but R-MDMA will not provide the experience these dutch mega beans give. As others have said before, R-MDMA is the longer lasting of the two not shorter. It is more a chill psychedelic-esque MDMA vibe that probably lasts about 6-8 hours. It is the isomer that gives you that chill sorta rolling feel once the S-isomer wears off around 3-5 hours after dosing. When I was younger I used to think certain batches contained more R or S isomer as a few batches I’d come across in my early days would lack the intense stimulation and euphoria, and were more psychedelic (not MDA psychedelic mind you) and longer lasting with less up and down. I’ve since concluded though that it may have been due to impurities as these types of experiences always were off of browner more rock like MDMA (indicative of impurities compared to actual crystal) with obvious impurities. While how I can understand my past assumptions I can’t understand how we can fit the profile of this new MDMA with that of the R-isomer.
Short acting effects, stimulation, shitty next day comedown don’t match.. And I doubt even a partially skewed enatiometric excess would give such effects, unless perhaps it was skewed towards the S-isomer. But even THEN we’d be seeing positive not negative effects. If either isomer gave all these negative effects you’d feel it even in a racemic mixture and it would drive us to produce enatiometrically pure product to remove the negative isomer. This has never been the case and likely isn’t now either. I’m sorry to those that have worked so hard on that theory but it’s time to move on.
While isomers is likely not the reason for our variation in todays MDMA, it is possible that it did have some effect on the product of the 90’s. Before I go into this I have no actual research to back up this claim, just a word from a reputable source and all the connections that can be made beyond this. After being told this however, I am leaning more towards this being the culprit for early 90’s bliss instead of different synthetic route like I outline above.
I’ve been told that during the late 80’s and early 90’s much of the MDMA being used had been produced as the pure S-isomer from labs in India and China while it was still legal, and in VERY LARGE amounts. This would make sense as at the time it was legal and likely MUCH cheaper to produce so discarding half of the end product wasn’t the biggest deal in the world. These large batches were then used until they dried up in the early to mid 90’s.
Now this actually starts to make sense when you think about it all. It is a fact that people claimed back then 75mg or so of MDMA would have you rolling for a solid 4-5 hours. As we know today this just doesn’t make sense with our typical racemic MDMA which produces an experience like that only in the 100-120mg range. Well looking at a graph that outlines the relationship between dosage and effect for R-MDMA, S-MDMA, and racemic MDMA, it seems a dosage in the 60-80mg range is equal to a racemic dosage of 100-130mg. This matches perfectly with the variation in dosage between then and now. 75mg of S-MDMA will have you floored out like you took 130mg+ pure racemic MDMA. (And to those that think 130mg of pure racemic isn’t going to "floor you".. I’d argue you just haven’t done PURE MDMA before.)
Next it seems the MDMA from back then is always referenced as being “speedier” and more “dancey” than the stuff these days. Well again this fits perfectly with the S-isomer which is definitely the speedier of the two when looking at both MDMA and MDA. It was this S-isomer that gave people that drive to dance, and the chill somewhat trippy aspect of the R-isomer wasn’t there to balance the effects.
Many reading this are probably thinking of the study in which participants ultimately found the racemic to be better than either isomer alone. I’d argue in ways that study can’t be completely relied on to make our assessments about exactly how these drugs should feel. It is likely the participants for that study were more familiar, fond and comfortable with psychedelic drugs that stimulants. So they likely found the balanced nature and slight psychedelia of the racemic to be superior to the highly stimulating S-isomer.
But for dancing your heart out all night and chatting up the randoms next to you, the R-isomer isn’t really “needed.” And so when all of the people who essentially birthed the rave scene went from consuming a more stimulating more potent product to the chiller less potent racemic, they couldn’t help but feel something was amiss. Of course racemic MDMA can do all of the things I just listed, like falling in love with the stranger next to you but it likely is missing that overwhelming urge to DANCE, DANCE, DANCE! When we compare to the two I’m sure..
Up next on the minor factors list I’ve got Salts. This was discussed as a possible explanation for the differences between MDMA batches back in the 00’s on BL here, but ultimately seemed to get thrown to the side for good reason. While salts do vary in MDMA batches, they likely don’t enough to give the huge differences we see in various locations. Above I talked about a Chinese paper that did analysis of many ecstasy tablets. They found one manufacturer in particular who they seized quite a few pills from, that produced the phosphate salt of MDMA (the dihydrogen phosphate salt in particular). This salt was found in 31 cases containing 292 tablets so it’s obvious this tablet wasn’t some small local boutique press. The MDMA freebase content is 66.3% for the dihydrogen phosphate salt, quite a bit less potent than MDMA HCl.
While many would argue that changing the salt will only effect bioavailability and dosage, it’s obvious from underground research by MDMA chemists that the salts can change more than that. It appears online there has been a decent amount of talk about MDMA citrate. It seems the effect of MDMA citrate varies from the HCl even when an equal dosage (accounting for variations in salt molecular weight) is taken, and it has less of the in-your-face intensity but also less crash too. Based on my experience with other drugs and varying salt forms, I’d also say that it can change more than dosage.
Sadly there isn’t a ton of research in this field but my guess is that salts likely are only a minor factor when looking at the picture as a whole. Not enough manufacturers change the salt up to make a huge difference that is seen worldwide.
And last but not least, binders… So many people think this has little effect but that isn’t not true in the slightest, and with a drug like MDMA the binders can greatly change the pharmacokinetics. Many of these Dutch Super Beans are very professionally pressed these days, with enteric coating and the whole 9 yards. Some people report not feeling effects for up to or over two hours!! With that being the case it’s in essence causing a slower timed release compared to MDMA in a capsule or a crumbly local press. MDMA is a fickle one when it comes to dosing, as soon as you take an initial dosage your enzymes begin to change in response.
Taking MDMA all at once is ideal because there is more MDMA to make it to the brain, but when taken in smaller redoses the enzymes begin to change and it is shown there is more MDMA to MDA metabolism along with more neurotoxicity overall. Taking a pill that causes a slow release will do both of these things as well, so expect perhaps a slightly more MDA-esque experience as well as a shittier comedown in the days following compared to dosing the whole thing quickly via crushed tablet or powder in a capsule.
This should be taken as note to people who take lots of small redoses throughout a night. You’re causing yourself more damage for less effect, despite the rationale that taking smaller more frequent doses would do the opposite and be less damaging.
Well that’s it for now folks, I’ll add more in later I just am so busy these days it’s hard but this topic is so complex this is just scraping the surface in my opinion. But there you have it, my theories backed with evidence as to why I believe MDMA batches and experiences vary.
-GC
Last edited: