-
Psychedelic Drugs Welcome Guest
PD's Best Threads Index Social ThreadSupport Bluelight Psychedelic Beginner's FAQ -
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
4-aco-dmt redundancies
- Thread starter yoyoman
- Start date
Xorkoth
Bluelight Crew
I'll be adding a report or two sometime soon. I'll definitely let you know.
Sprinklervibes
Bluelighter
Uhm, regarding this blue colour and hydrolysis. I had mushrooms which I wanted to make a tea out of(without heating). I cut them up good and placed them in a bottle of grapefruit juice. The blue spots on the mushrooms dissapeared in a short period. I drank half of the dose at a festival, and took the other half back home. A few days after doing the "extraction", the juice had totally become blue/black. What is the cause of this? Does it have to see with mushrooms turning blue, too?
Sprinklervibes
Bluelighter
So adding more vitamine C or other acid(I don't think grapejuice is so acidic due to vit c) would bring the molecule back to the original state?
Also, what does it oxidizes into? Is it still active?
Also, what does it oxidizes into? Is it still active?
Ok, well I put ~30mg 4-aco-dmt fumarate in a small clear glass thing with distilled water (not that much water) + NaOH added (about 20mg NaOH). Swirled it around / made sure it was all dissolved.
I lit an alcohol lamp for a heat source.. well when I went to look to pick up the container with the 4-aco-dmt in it, it was already turning to a very light blue. Well I added a little heat anyway.. it seemed to just turn to a darker blue then went a little bit greenish (bluegreen i guess). So I just dumped the bluegreen liquid into a cup, added some acetic juice to it and drank it down
------
Kicked in within 15mins after i drank it. It does feel more like a 'pure' trip, at +0.40mins ... well lol what else can i say? i prefer this kind of trip over the 4-acetoxy-dmt just the differences are hard to put into words the visuals are more like mushrooms...head feels more like mushrooms
the visuals are more like mushrooms...head feels more like mushrooms
... more DMT like fluidity here.. colors more *brighter* in the way mushrooms do it wheres 4-ace-dmt they .. well its just "different" hehe..
I think some - not much though - was oxidised to a non-goodygoody product when it went darker blue / greenish blue w/heat..
well uh.. off the computy i go.
I lit an alcohol lamp for a heat source.. well when I went to look to pick up the container with the 4-aco-dmt in it, it was already turning to a very light blue. Well I added a little heat anyway.. it seemed to just turn to a darker blue then went a little bit greenish (bluegreen i guess). So I just dumped the bluegreen liquid into a cup, added some acetic juice to it and drank it down
------
Kicked in within 15mins after i drank it. It does feel more like a 'pure' trip, at +0.40mins ... well lol what else can i say? i prefer this kind of trip over the 4-acetoxy-dmt just the differences are hard to put into words
... more DMT like fluidity here.. colors more *brighter* in the way mushrooms do it wheres 4-ace-dmt they .. well its just "different" hehe..
I think some - not much though - was oxidised to a non-goodygoody product when it went darker blue / greenish blue w/heat..
well uh.. off the computy i go.
oxidation
Yepp!
Quod erat demonstrandum. I’m glad, that my suggestions verified.
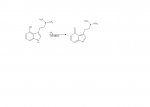
I attached the formulae of the free phenol and its quinonic oxidation product. I think the drawn oxidation product might be also active, since it might be reduced again in the body by enzymes.
The problem is the chemical reactivity of such quinones. Their strong electrophilic properties make them easily react with itself to form (most probably inactive) polymers or with any present nucleophil.
Yepp!
Quod erat demonstrandum. I’m glad, that my suggestions verified.
I attached the formulae of the free phenol and its quinonic oxidation product. I think the drawn oxidation product might be also active, since it might be reduced again in the body by enzymes.
The problem is the chemical reactivity of such quinones. Their strong electrophilic properties make them easily react with itself to form (most probably inactive) polymers or with any present nucleophil.
Attachments
yeah this entire trip is different than aco-dmt, and its changing, i think that oxidation product is active, like right now colors are extra vibrant.. visuals are better.. super clear vision..
maybe if i tried that next time but let it get to a real deep color?
maybe if i tried that next time but let it get to a real deep color?
nuke
Bluelighter
- Joined
- Nov 7, 2004
- Messages
- 4,191
EN21 said:Yepp!
Quod erat demonstrandum. I’m glad, that my suggestions verified.
I attached the formulae of the free phenol and its quinonic oxidation product. I think the drawn oxidation product might be also active, since it might be reduced again in the body by enzymes.
The problem is the chemical reactivity of such quinones. Their strong electrophilic properties make them easily react with itself to form (most probably inactive) polymers or with any present nucleophil.
that's a ketone, not quinone.
orbital_forest
Bluelighter
well,i think im going to have to pick a little of this stuff up. were your doses with the fumarate BD?
nanobrain
Bluelighter
somewhere on the internet i read...bigup props to the DMT elves..
Trogdor
Bluelighter
Quageschi
Bluelighter
- Joined
- Apr 2, 2006
- Messages
- 108
Despite all my studying of drugs i seem to have never picked up on the fact that Psilocin is so closely related to DMT (chemicaly of course). So this leaves me with a few questions that i hope one of you can answer. First is what is it molecularly that seperates DMT and psilosin (in the pictures it looks like OH, in the chemical formula it looks like just O). My second question is what is psilocin's shortenend chemcal name (4-Hydroxy-N,N-dimethyl-tryptamine), 4-OH-DMT? What about psilocybin? 4-OP-DMT? Sry this is kind of off topic
Morninggloryseed
Bluelight Crew
psilocin is 4-Ho-DMT and psilocybin is 4-Po-DMT or '4-Ho-DMT, phosphate ester'.
nuke
Bluelighter
- Joined
- Nov 7, 2004
- Messages
- 4,191
4-acetoxy-dmt mule said:Summary: This is indeed much like a pure shot of psilocin, however the visuals lack the depth that I suspect is added by the phosphate ester contained by natural cubensis.
Well, since the 4-PO is too polar to make it into the brain, I reckon all the effects of mushrooms are from 4-HO-DMT. The 4-PO probably just slows the intake of 4-HO-DMT (as it has to convert before it can go into your brain).
And, yeah, in my experience, 4-HO-Ts are more visual than 4-AcO-Ts, even when accounting weight. If the 4-AcO-T is active in and of itself (probably), then perhaps it has more avisual effects similar to say 4-MeO-MiPT rather than the hydroxylated counterparts.
4-acetoxy-dmt mule
Greenlighter
- Joined
- Sep 1, 2006
- Messages
- 15
nuke said:words.
I simply don't know, and as I am only a relative beginner in the chemistry side, I cannot speculate very far. I just know the subjective difference, as I have experienced pure 4-HO-DMT before and this is not the same thing. Nor is it the same as the combination of 4-HO/4-PO/baeocystin (4-??) that is found in cubensis. It's a new experience, but of course one that compares well to shrooms.
Is there an easy way to do a home conversion of 4-ACO-DMT to 4-HO-DMT? I remember a shroomery discussion about acidic juices such as lemon and grapefruit doing the psilocybin->psilocin conversion in the glass before ingestion, perhaps this would work for the acetate ester as well?
Of course I'm probably way, way off and this is not chemically possible at all.
blue)dolphin
Bluelighter
- Joined
- May 14, 2006
- Messages
- 346
I find mushroom / psilacetin / DMT visuals to be pretty similar.
Of course it takes a lot of shrooms to get visuals like a 30-40mg DMT dose (smoked)!
But say, 15mg DMT in a pipe makes things look fairly similar to, say, 10mg psilacetin to me. Doses may vary... but assuming you trip equally hard on both substances, the visuals are very similar to me.
However, since DMT has such a rapid onset, rush, and strong vibrating buzz, DMT visuals tend to act more quickly and be a bit more flashy. Psilacetin comes on slow, relaxes the user (in my experience and others I know) and therefore the visuals seem more relaxed -- they come at you at a pace that is very easy to accept and absorb.
To me this is a huge advantage, and one reason I consider both psilacetin and mushrooms to be better psychedelics than DMT.
In fact, sitting in a car peaking on 10mg of psilacetin with the rain falling on the windsheild was a beautiful relaxing experience. When a car drove by it looked like somebody took a paint bucket full of light and tossed it over my surroundings.
Walking along an empty dirt road at night, surrounded by large trees, I certainly felt a strong connectedness to everything around me. It reminded me very much of mushrooms, but I felt clearer headed for the level of trip I was experiencing. The occasional house did indeed look otherworldly, and the world around me took on the look and feel of something out of a fairy tale.
Quite magical stuff, I'd say. I consider myself very lucky to have had the opportunity to try this one.
For me, DMT, LSD, mushrooms, and psilacetin all have fantastic visuals... and unless I'm forgetting something, these are the best I've tried visually. However, DMT is almost useless for me because it comes on too quickly, grabs you by the balls, and its hard to integrate. LSD is too long for most occasions and is usually not a very "relaxing" experience. Great fun once in a while, but certainly not "all purpose". Mushrooms are fantastic, but leave my stomach feeling like shit half the time and are hard to dose accurately with.
That leaves psilacetin as my favorite all time psychedelic! To me it's equal to shrooms but you can dose it *exactly* and there are no significant negative physical side effects on me and the few people I know who've also tried it.
proceed with caution. And stock up while you still can! I'm certainly not holding my breath for anything better than *this* to come along any time soon
Of course it takes a lot of shrooms to get visuals like a 30-40mg DMT dose (smoked)!
But say, 15mg DMT in a pipe makes things look fairly similar to, say, 10mg psilacetin to me. Doses may vary... but assuming you trip equally hard on both substances, the visuals are very similar to me.
However, since DMT has such a rapid onset, rush, and strong vibrating buzz, DMT visuals tend to act more quickly and be a bit more flashy. Psilacetin comes on slow, relaxes the user (in my experience and others I know) and therefore the visuals seem more relaxed -- they come at you at a pace that is very easy to accept and absorb.
To me this is a huge advantage, and one reason I consider both psilacetin and mushrooms to be better psychedelics than DMT.
In fact, sitting in a car peaking on 10mg of psilacetin with the rain falling on the windsheild was a beautiful relaxing experience. When a car drove by it looked like somebody took a paint bucket full of light and tossed it over my surroundings.
Walking along an empty dirt road at night, surrounded by large trees, I certainly felt a strong connectedness to everything around me. It reminded me very much of mushrooms, but I felt clearer headed for the level of trip I was experiencing. The occasional house did indeed look otherworldly, and the world around me took on the look and feel of something out of a fairy tale.
Quite magical stuff, I'd say. I consider myself very lucky to have had the opportunity to try this one.
For me, DMT, LSD, mushrooms, and psilacetin all have fantastic visuals... and unless I'm forgetting something, these are the best I've tried visually. However, DMT is almost useless for me because it comes on too quickly, grabs you by the balls, and its hard to integrate. LSD is too long for most occasions and is usually not a very "relaxing" experience. Great fun once in a while, but certainly not "all purpose". Mushrooms are fantastic, but leave my stomach feeling like shit half the time and are hard to dose accurately with.
That leaves psilacetin as my favorite all time psychedelic! To me it's equal to shrooms but you can dose it *exactly* and there are no significant negative physical side effects on me and the few people I know who've also tried it.
proceed with caution. And stock up while you still can! I'm certainly not holding my breath for anything better than *this* to come along any time soon
nuke
Bluelighter
- Joined
- Nov 7, 2004
- Messages
- 4,191
4-acetoxy-dmt mule said:I simply don't know, and as I am only a relative beginner in the chemistry side, I cannot speculate very far. I just know the subjective difference, as I have experienced pure 4-HO-DMT before and this is not the same thing. Nor is it the same as the combination of 4-HO/4-PO/baeocystin (4-??) that is found in cubensis. It's a new experience, but of course one that compares well to shrooms.
Is there an easy way to do a home conversion of 4-ACO-DMT to 4-HO-DMT? I remember a shroomery discussion about acidic juices such as lemon and grapefruit doing the psilocybin->psilocin conversion in the glass before ingestion, perhaps this would work for the acetate ester as well?
Of course I'm probably way, way off and this is not chemically possible at all.
You've had pure 4-HO-DMT before? I'm curious, where on Earth did it come from (however vague)?
As for the conversion to 4-HO-DMT (is this synthesis discussion?), you should be able to get by just by adding a small amount of a moderately strengthed base and heating in h2o until the colour of your solution starts to blue or green a little. That signals the formation of the degredated ketone psilocinone. You might have to add a little more than enough to salt with the separated acetic acid that might form when the acetyl breaks free from the tryptamine (this is speculative and not entirely confirmed by testing).
Last edited: