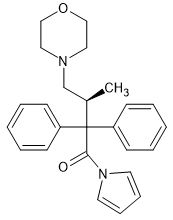
I have to admit that a pyrrolidine can adopt 2 conformers for it's minimum energy conformation and although their are not chiral, steric bulk may prevent the C-N bond from rotating. I am still quite sure that the pyrrole homologue would be almost identical to dextromoramide. In fact, duration might be slightly longer.
Image dm hosted on ImgBB

ibb.co
The pyrrole ring will be planer whereas the pyrrolidine ring has bond angles of about 109° so it isn't planer. Now, I admit that this is a total guess, but I do not think that this makes much difference to affinity or activity. As far as I know, Janssen didn't try placing aromatics on amide, ketone or ester moieties (but maybe someone else knows better). There are quite a few cases where he COULD, but he didn't.
I think that the reason that a 5-membered ring is the only homologue that is highly active is that it's space-filling (in conjunction with that alpha methyl) rotates one of the benzene rings so that it fits the receptor, affinity is increased a great deal. I mean, the piperidine homologue is almost inactive. Of course, he never tried 2,3-dimethyl cyclopropane or 2,3-cyclobutane homologues.. Imagine - lots of fun with optical isomers!
But it is my experience that when medicinal chemists discover a REALLY active homologue, they tend to take the afternoon off. I know this sounds glib but Dan (Lednicer) never made the p-Me homologue of BDPC and when I asked 'why?' he just said 'in all of the excitement, we forgot.'. I mean, I LOVE Dan for that, he was such a human person.
I guess the other (in)famous example of space-filling is when the diethylamide moiety of LSD can be replaced by an (S,S)2,4-dimethylazetidine or a mono sec-butylamide. Hoffman found this example that was magnitudes more potent than closely related compounds and just stopped. I guess from a medicine chemists point of view, they HAVE an appropriate ligand and their would be no prizes for finding a second one.
For this interested, in the N-benzyl derivatives of the PEA class, the benzene ring fills the space occupied by the diethyl in LSD and the O of the -OH or -OCH overlays the =O of the amide moiety.
I mean, I respect the German guy for thinking outside the box and for proving that an o-hydroxy or o-methoxy benzene acts as a bioisostere to a diethylamide.
I seem to have rambles.... sorry.