sekio
Bluelight Crew
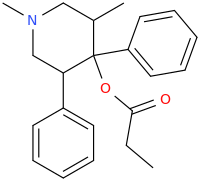
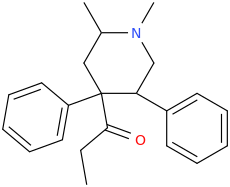
Cyclic analogs of propoxyphene / methadone as substituted pethidine / ketobemidone / prodine analogs
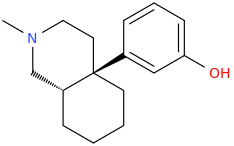
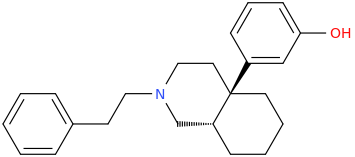
US4219652A compounds, opioid analgesics
![1,2,3,4,5,6,7-heptahydro-N-methyl-9-hydroxy-Benzo[b]isoquinolino[4a,4-d]furan.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F1%2C2%2C3%2C4%2C5%2C6%2C7-heptahydro-N-methyl-9-hydroxy-Benzo%5Bb%5Disoquinolino%5B4a%2C4-d%5Dfuran.png&hash=d8184af3fdc0b3992a83dd1e05eca343)
Related
![1,2,3,4,5,6,7-heptahydro-N-methyl-2-oxo-4a,9-dihydroxy-12-methyl-Benzo[b]isoquinolino[4a,4-d]furan.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F1%2C2%2C3%2C4%2C5%2C6%2C7-heptahydro-N-methyl-2-oxo-4a%2C9-dihydroxy-12-methyl-Benzo%5Bb%5Disoquinolino%5B4a%2C4-d%5Dfuran.png&hash=8a2156da7dc7e77e6d0262444412254a)
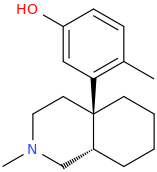
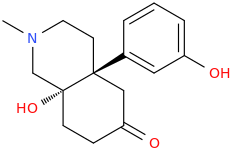
9,10-seco-oxymorphone
Last edited:
N&PD Moderators: Skorpio
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
I Like to Draw Pictures of Random Molecules
sekio
Bluelight Crew


Cyclic analogs of propoxyphene / methadone as substituted pethidine / ketobemidone / prodine analogs


US4219652A compounds, opioid analgesics
![1,2,3,4,5,6,7-heptahydro-N-methyl-9-hydroxy-Benzo[b]isoquinolino[4a,4-d]furan.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F1%2C2%2C3%2C4%2C5%2C6%2C7-heptahydro-N-methyl-9-hydroxy-Benzo%5Bb%5Disoquinolino%5B4a%2C4-d%5Dfuran.png&hash=d8184af3fdc0b3992a83dd1e05eca343)


Related
![1,2,3,4,5,6,7-heptahydro-N-methyl-2-oxo-4a,9-dihydroxy-12-methyl-Benzo[b]isoquinolino[4a,4-d]furan.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F1%2C2%2C3%2C4%2C5%2C6%2C7-heptahydro-N-methyl-2-oxo-4a%2C9-dihydroxy-12-methyl-Benzo%5Bb%5Disoquinolino%5B4a%2C4-d%5Dfuran.png&hash=8a2156da7dc7e77e6d0262444412254a)
9,10-seco-oxymorphone
Last edited:
@ekio the ones from the third one are looking a bit similar to mesembrine, mesembrenone and similar alkaloids from kanna, I guess they could have serotonergic activity?
---
some (very) random chems: (It would be great to have a computer program that could analyse the structure to give you all the infos like possible binding sites, probable hydrolysis-parts, most similar known molecules etc. I use Marvin Sketch from ChemAxon (it's a free and really good software) but it can only give very general analysis data like melting point, elemental mass etc.)
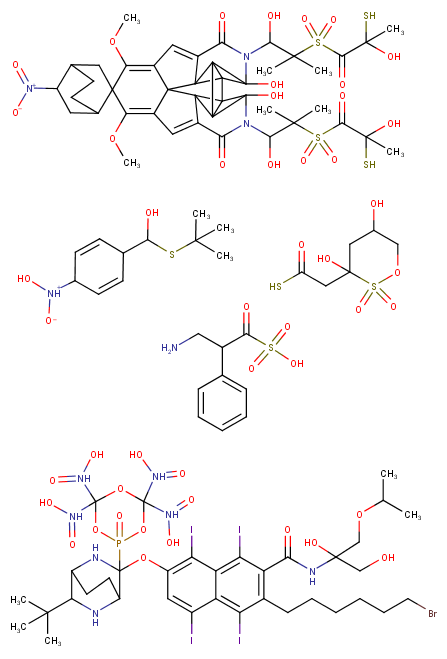 Please, fix your nitro groups
Please, fix your nitro groups
![]()
Nitro groups have the nitrogen accept a positive charge, with a double bond to one oxygen, and a single bond to the other oxygen, which carries a negative charge. That way, all of the atoms achieve a full octet of electrons in their valence shell. There are no protons in nitro groups, neither on the oxygen nor on the nitrogen.
Also, you generally wouldn't want to put that many electron-withdrawing groups on a single carbon, as these tend to readily hydrolyze in acidic conditions.
Deleted member 170540
Bluelight Crew
^ Wouldn't that (the compound above) be hydrolyzed immediately like most acid anhydrides?
sekio
Bluelight Crew
yes, it'd quickly hydrolize to the "pethidinic acid" equivalent + acetic acid and would therefore be inactive, better to leave it as an ester or ketone than make it an acid anhydride...
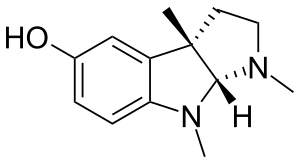
eseroline, metabolite of physostygmine, is apparently an opioid agonist but is also neurotoxic at uM levels. (apparently) ... i hypothesize that if the Ki for interaction at the opioid receptors is in the nM range that it would still be an interesting cmpd to try once or twice
Deleted member 170540
Bluelight Crew
Quite interesting, as it would be easy to make by hydrolyzing the physostigmine extracted from the seeds of the plant that produces it. Probably would be best to dose it with some atropine or scopolamine to prevent cholinergic side effects.
^ Wouldn't that (the compound above) be hydrolyzed immediately like most acid anhydrides?
Not only that, but it would basically be impossible to make, since any attempt at acetylating the carboxyl group (to make the anhydride) would first acetylate the phenolic OH group.
Sure, you could try using a protecting group on the phenol to protect it during the acetylation step, but how are you going to remove that protecting group without also cleaving the anhydride?
sekio
Bluelight Crew
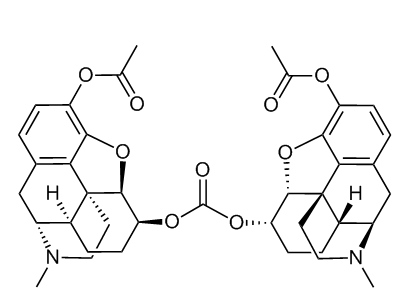
from US20050154002A1, carbonate ester prodrug of 3-Ac-dihydromorphine
Apparently this is suitable for transdermal applications!

from US20050154002A1, carbonate ester prodrug of 3-Ac-dihydromorphine
This brings up an interesting legal question: is the dimer of a compound an analogue of it? That would be an interesting case to see!
sekio
Bluelight Crew
I would think it is, but that's just me. Especially because you'd expect to see it hydrolyse fairky easily to the parent cmpd.
This brings up an interesting legal question: is the dimer of a compound an analogue of it? That would be an interesting case to see!
The UN Single Convention on Narcotic Drugs specifically mentions that the various isomers, salts, ethers and esters of schedule I drugs are to be put in the same schedule by default, unless specified otherwise (ex.: DXM is a stereoisomer and ether of the potent opioid levorphanol, but is specifically excluded from schedule I due to the lack of opioid activity).
When national governments and international regulatory bodies around the world were passing laws regulating the sale of heroin, some of the companies producing it were quick to switch to other morphine esters, such as benzoylmorphine or propionylmorphine. In 1929, German pharm companies were reported to be converting around 19 tons of morphine into then-uncontrolled esters and ethers of morphine. After Germany passed ze Opiumgesetz, that amount dropped to just ~4 tons within two years, most of which was codeine and its homologue ethylmorphine, representing the actual medicinal demand for these substances.
Sekio's dimer is really just a fancy ester (3'-acetyldihydromorphinylcarbonyl-3-acetyldihydromorphinate?), if you think about it, and as such it would probably be subject to UN regulations, not to mention various blanket bans, analogue acts and opium laws at the national level.
sekio
Bluelight Crew
Technically speaking, it's an ester of carbonic acid. I'd call it bis(3-acetyldihydromorphinyl)carbonate.
I wonder if malonic, succinic, glutaric, adipic acid or others would work too. Terephthalic acid? Fumaric/maleic acid?
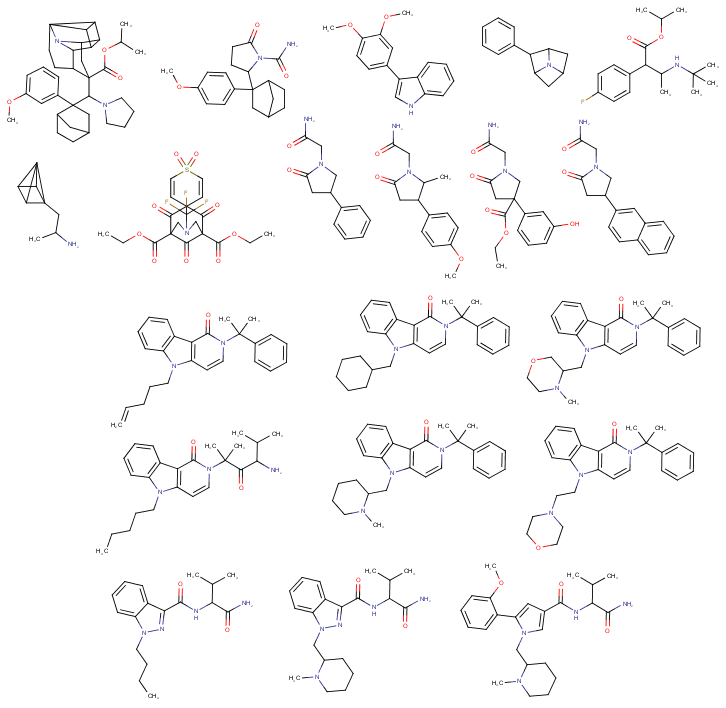
Can anyone explain where the difference between a pentyl-chain like usually found in cannabinoids and a pentene-yl chain like you can find in some of today's overly potent cannabinoids (thanks governments for "protecting" people so well through laws...). I know that some metabolites of many cannas like JWH122-pentenyl exist but if they can be effective (like that overly potent one 5-CL-ADB-A) why aren't they more widely used? Seems like a simple change in chemistry design. (For example there is a pentene-yl chain on the third cannbinoid above the bottom left one)
Last edited:
sekio
Bluelight Crew
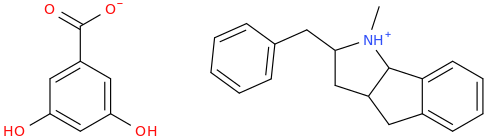
(+-)-HBMP resorcylate, formerly called (+-)-RTI-4793-14, ligand for "PCP site 2" & monoamine uptake inhibitor
simply put, the pentenyl chain 'looks' a lot like the pentyl chain, and is still nonreactive enough that it doesn't mess with the drug's binding or half life by too much.
Can anyone explain where the difference between a pentyl-chain like usually found in cannabinoids and a pentene-yl chain like you can find in some of today's overly potent cannabinoids
Last edited:
why aren't they more widely used? Seems like a simple change in chemistry design. (For example there is a pentene-yl chain on the third cannbinoid above the bottom left one)
While alkanes are based on a tetrahedral geometry (as in: if your carbon atom is in the center of a tetrahedron, then the 4 atoms it is bonded to are located in the corners), alkenes have a planar (i.e. flat) geometry.
Additionally, there is little to impede the rotation of a single bond, whereas the double bond of an alkene is much stiffer.
This means that the actual 3-dimensional structure of the molecule, and thus the way it interacts with receptors, can be slightly different.
However, the problem with alkenes is that these "unsaturated" hydrocarbons are generally more reactive than their saturated cousins. For example, exposure to acids like HCl can result in hydrohalogenations (turning the alkene into a saturated haloalkane); exposure to Br2 or Cl2 will turn them into alkanes substituted with two halogens; oxidants can cause the alkene to be converted into a highly reactive epoxide, or to be cleaved apart.
This can sometimes complicate the synthesis of a compound, or result in poor storage-lives or biological half-lives, and possibly even lead to the formation of toxic metabolites.
Last edited:
For example, exposure to acids like HCl can result in hydrohalogenations (turning the alkene into a saturated haloalkane)
Actually, looking at the structures of 5-Cl-ADB-A and comparing it to that of regular 5-Cl-ADB, it looks like what they did was the opposite of the reaction mentioned above: They first made the compound with a chloropentyl chain on it, then treated it with a very strong base in order to achieve a dehydrohalogenation, i.e. the removal of HX (X = any halogen) from a haloalkane to turn it into an alkene.
Still, even if you only did it at the very end, that's an extra reaction step compared to regular 5-Cl-ADB... and there's always the risk of side-reactions (although these can usually be minimized by using a very bulky base that can attack the protons on the exposed alkyl chain of the molecule, but not the carbonyls of the ester and amide groups).
sekio
Bluelight Crew
most hydrohalogenations need to be run with anhydrous HCl to be effective as the covalent species H-Cl is not present in a high enough concentration in aqueous solutions (being a strong acid, it dissociates completely to H₃O+ and Cl-)
alkenes are actually pretty unreactive, save for some exotic conditions (MnO4-, OsO4, dihalogens, Hg2+, ozone, Pd/O2 (wacker))