-
N&PD Moderators: Skorpio | thegreenhand
-
Neuroscience & Pharmacology Discussion Welcome Guest
Posting Rules Bluelight Rules Recent Journal Articles Chemistry Mega-Thread FREE Chemistry Databases! Self-Education Guide
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Dresden's Chemical Fluff Thread (Name-A-Molecule)
- Thread starter Dresden
- Start date
Ketamania
Bluelight Crew
- Joined
- Dec 30, 2017
- Messages
- 1,120
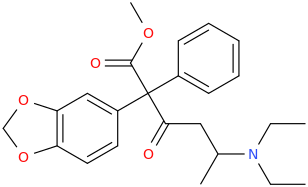
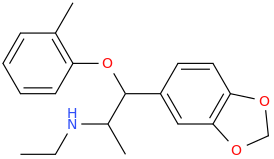
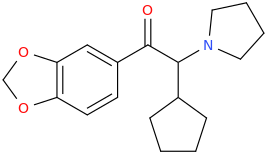
a-PCYP
2-cyclohexyl-1-phenyl-2-(pyrrolidin-1-yl)ethan-1-one
A cathinone (bath salt) that I have experimented upon. Fantastic high, seriously high heart rate. Way more euphoric than any stimulant i have ever tried, better than meth. However, it is EXTREMELY addicting and ended up having to flush this one. I can see how one can have a heart attack if they take took much or are susceptible to cardiac problems. I'd rate the high 10/10 but the comedown a -100/10. Overall, don't try it. It won't make you into a 'zombie' or anything, giant myth.
2-cyclohexyl-1-phenyl-2-(pyrrolidin-1-yl)ethan-1-one

A cathinone (bath salt) that I have experimented upon. Fantastic high, seriously high heart rate. Way more euphoric than any stimulant i have ever tried, better than meth. However, it is EXTREMELY addicting and ended up having to flush this one. I can see how one can have a heart attack if they take took much or are susceptible to cardiac problems. I'd rate the high 10/10 but the comedown a -100/10. Overall, don't try it. It won't make you into a 'zombie' or anything, giant myth.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,439
Your Pharmies Are Basic Compared To Mine. A Search For The Higher Intelligence. The Most Benevolent King. Communicating Through Your Dreams. Understanding Makes My Truth Crystal Clear.
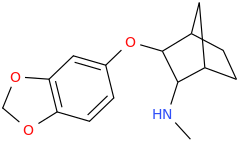
PLOYD FENNINGTON
1-(3,4-methylenedioxyphenyl)-1-oxo-2-(1-pyrrolidinyl)-2-cyclopentylethane

PLOYD FENNINGTON
1-(3,4-methylenedioxyphenyl)-1-oxo-2-(1-pyrrolidinyl)-2-cyclopentylethane
Last edited:
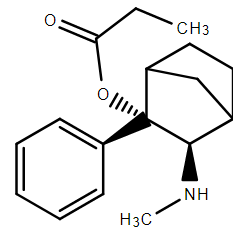
And just for a change, here's one that is known to work. The methylene bridge places the N: in the same relative position to the aryl as the aryl:methylamine of nortilidine. Of course, it's fun, fun, fun to make but it IS very rigid and so it's affinity is pretty decent. I cam across it in a 1960s German paper on tilidine and related compounds. Always remember that it isn't the N but the N: position & orientation is what matters. That is why unusual compounds like dezocine are primary amines and why they are mixed agonist/antagonist.
MANY years ago researchers produced quaternary salts of common opiates like morphine so, in practice there was a chiral centre and it turned out that 1 isomers was an agonist, the other was either an antagonist (phenol or bioisostere thereof) allowing.
I find it interesting because it showed that some keen medicinal chemists asked a simple 'what if?' and proved it to be true.
Secondary amines usually have a conformation that places the N-alkyl opposing the N: and so they turn out to be agonists. Above you will note an example of a secondary amine. I suspect it is a total bitch to make and some poor post-grad student ended up as an 'et al' even if it took them a year.
You will note that like tramadol (also a DRI/NRI without the ester) is closely related. I think I have pointed out that it is possible to produce a highly EUPHORIC opioid (due to DRI action) based on some quite average DRI/NRI lligands.
Since lat post I've had the chance to try the N-methyl homologue of isopilidine. It lasts for a good 6 hours. It is highly euphoric so all I can say is that people do NOT try to find a regular dealer. I've seen chemist/users who were obtaining bottles of the stuff and converting it to something very similar,just x10 more potent.
Of course, it's the DRI/NRI action that makes it different BUT I do not know IF it has NMDA activity, someone could make billions. Now, think about that - if dealers make billions, WHO LOSES.

Nowm, I admit that I have suggested MANY terminal aryls (based on Lednicer's work) but while not being the compute, Lednicer mentioned this as a more practical, longer-acting analogue. The KEY is that usually the aromatic is hydroxylated but with many 5-membered heterocycles, this isn't possible. The 2-thiophene analogue was known and tested and turned out to be x267 (rather than x204 for the parent.
Of course, the key is NOT to rely on aromatic hydroxylation because it is not relaible between to patients. Yeah, we could make it VERY hard to metabolise, but something x504 REALLY does not need modification.
If you must play, swap the phenolic -OH of morphine and derivatives to a carboxmide which has a MUCH longer duration.
We may also do well to look at the methadone analogues, Several were noted as being 'moreish' i.e. people took for fun, tot for main.
Well, whatever, expect hydromorphone analoguse very soon. JUST ABOUT kitchen chemistry and fuck, it it's x10 H, EVERYONE will be at it.
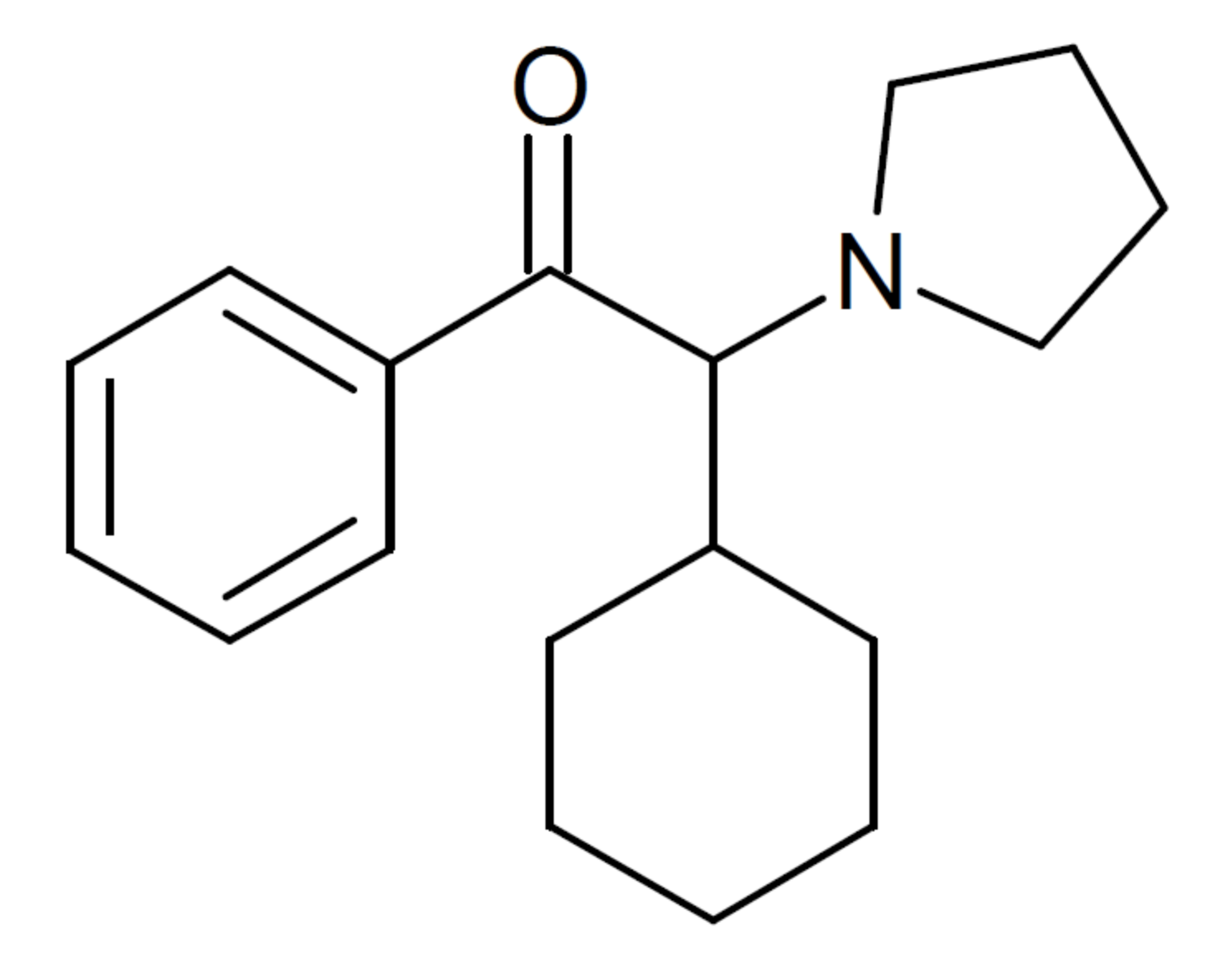
a-PCYP
2-cyclohexyl-1-phenyl-2-(pyrrolidin-1-yl)ethan-1-one

A cathinone (bath salt) that I have experimented upon. Fantastic high, seriously high heart rate. Way more euphoric than any stimulant i have ever tried, better than meth. However, it is EXTREMELY addicting and ended up having to flush this one. I can see how one can have a heart attack if they take took much or are susceptible to cardiac problems. I'd rate the high 10/10 but the comedown a -100/10. Overall, don't try it. It won't make you into a 'zombie' or anything, giant myth.
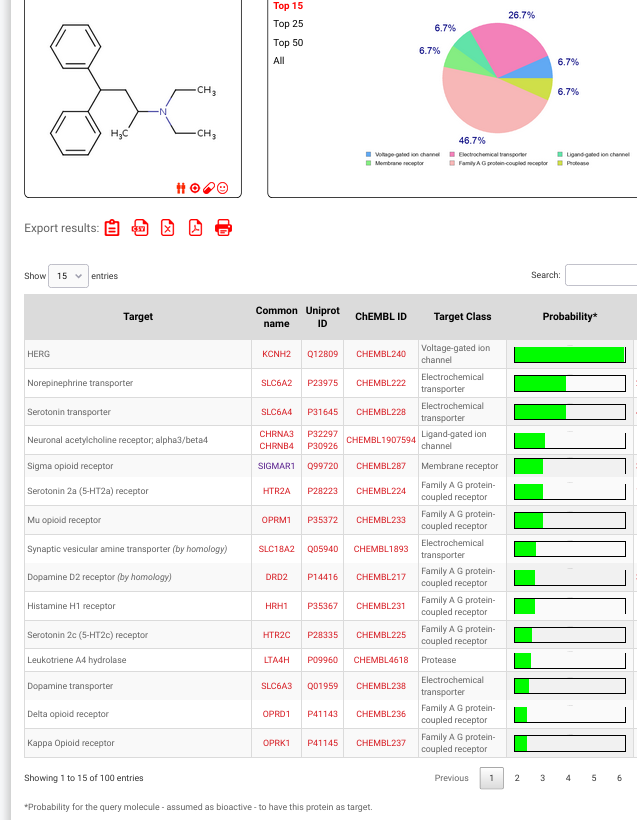
Made, stimulant. The 1,2-diary was more potent.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,439
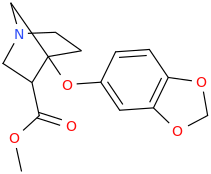
I'M WITH STOOPID
1-aza-3-carbomethoxy-4-(oxapiperonyl)norbornane
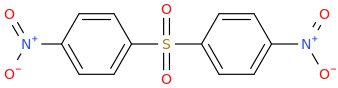
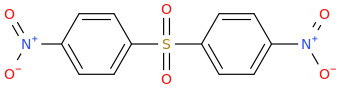
NITRODAPSONE
di(4-nitrophenyl)sulfone
broad spectrum antimicrobial for acne, Leprosy (Lou Hansen's disease), syphilis, pinta, yaws, Lyme's disease etc.
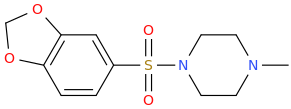
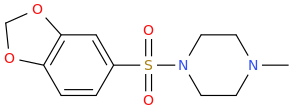
MIAGRA
1-methylpiperazin-4-yl 3,4-methylenedioxyphenyl sulfone
possible erection enhancer
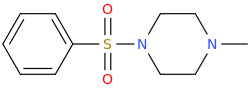
CON Q PISS ANT
1-methylpiperazin-4-yl phenyl sulfone
possible aphrodisiac
"Be Like The Ant."--The Bible.
Last edited by a moderator:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,439
![(2S,5R,6R)-2-carbomethoxy-3,3-dimethyl-7-oxo-6-(2-phenyl-1-methylethylamino)-4-propylsulfanyl-1-azabicyclo[3.2.0]heptane.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F%282S%2C5R%2C6R%29-2-carbomethoxy-3%2C3-dimethyl-7-oxo-6-%282-phenyl-1-methylethylamino%29-4-propylsulfanyl-1-azabicyclo%5B3.2.0%5Dheptane.png&hash=71b4680a62f740928570e8ea9106b6aa)
REESE STANFORD WITHERSPOON
(2S,5R,6R)-2-carbomethoxy-3,3-dimethyl-7-oxo-6-(2-phenyl-1-methylethylamino)-4-propylsulfanyl-1-azabicyclo [3.2.0]heptane
Many Things Are Possible When You Don't GAF Who Gets The Recognition
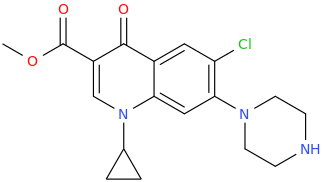
TAMISHA WHAT U TALKING ABOUT ARNOLD BRUCE WILLIS DEMI MOORE HOWARD STERN HUGHES LEONARDO DICAPRIO KIMBERLY TONY DANZA
3-carbomethoxy-1-cyclopropyl-6-chloro-4-oxo-7-(piperazin-1-yl)quinoline
antibiotic
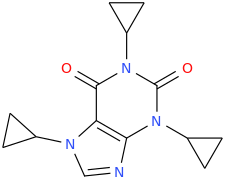
WHOOPI ARNOLD SISTER ACT KINDERGARTEN COP GOLDBERG SCHARZENEGGER
1,3,7-tricyclopropylxanthine
terra incognita
Cogito, ergo zoom.
![2-carbomethoxy-6-(2-(4-methoxyphenyl)-1-(methyl)ethylamino)-3-%7B2-[(iminoethyl)amino]ethyl%7Dthio-7-oxo-1-azabicyclo[3.2.0]hept-2-ene.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F2-carbomethoxy-6-%282-%284-methoxyphenyl%29-1-%28methyl%29ethylamino%29-3-%257B2-%5B%28iminoethyl%29amino%5Dethyl%257Dthio-7-oxo-1-azabicyclo%5B3.2.0%5Dhept-2-ene.png&hash=b5cf00bfa24cbb2a91fc1cbd1451f022)
FLORENCE NIGHTINGALE KERRY ZDRAVKOVICH SEARLE SHEILA NAOMI CAMPBELL WILLIAMSII CATENA THE 3RD
2-carbomethoxy-6-(2-(4-methoxyphenyl)-1-(methyl)ethylamino)-3-{2-[(iminoethyl)amino]ethyl}thio-7-oxo-1-azabicyclo[3.2.0]hept-2-ene
I told you so.
Last edited by a moderator:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,439
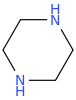
PIPERAZINE
anthelmintic
![(6S)-2-carbomethoxy-3-propylthio-6-(dichloroacetamido)-4,7-dioxo-1-azabicyclo[3.2.0]hept-2-ene.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F%286S%29-2-carbomethoxy-3-propylthio-6-%28dichloroacetamido%29-4%2C7-dioxo-1-azabicyclo%5B3.2.0%5Dhept-2-ene.png&hash=ca698014e65ad674a97f3fce3f7d3e93)
WHOOP! there it is.
2-carbomethoxy-3-propylthio-6-(dichloroacetamido)-4,7-dioxo-1-azabicyclo[3.2.0]hept-2-ene
I AM THE RESURRECTION.
![(6S)-2-carbomethoxy-6-%7B[(2R)-2-amino-2-(4-hydroxyphenyl)acetyl]amino%7D-4,7-dioxo-3-propylthio-1-azabicyclo[3.2.0]heptane.png](/community/proxy.php?image=https%3A%2F%2Fopsin.ch.cam.ac.uk%2Fopsin%2F+%286S%29-2-carbomethoxy-6-%257B%5B%282R%29-2-amino-2-%284-hydroxyphenyl%29acetyl%5Damino%257D-4%2C7-dioxo-3-propylthio-1-azabicyclo%5B3.2.0%5Dheptane.png&hash=477b90bd56ae1148c39acd5c22a85a79)
CHANDRAN AMOXICILLIN IMIPENEM HARVARD MIT SESHARGIRI
(6S)-2-carbomethoxy-6-{[(2R)-2-amino-2-(4-hydroxyphenyl)acetyl]amino}-4,7-dioxo-3-propylthio-1-azabicyclo[3.2.0]heptane

OM NAMA SHIVAYA
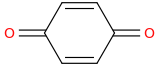
EMINEM
para-benzoquinone
Pd(II)Cl2
WENDY CRUMPLER FAEMS
CH3OH deadly
WOOD ALCOHOL
ETHYL ALCOHOL
CH3CH2OH
PROPYL ALCOHOL
CH3CH2CH2OH
Ethyl Tricks Methyl
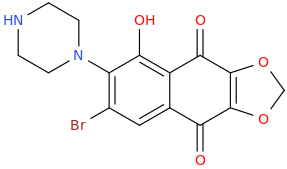
KENDRA MICHELLE OBAMA JASON JONES JENN ALEXANDER WARD
7-bromo-6-piperazinyl-2,3-methylenedioxy-5-hydroxy-1,4-naphthalenedione
Have A Disease?
There's A Cure For That!
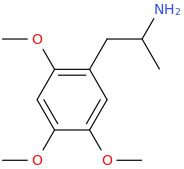
CALAMITY JANE
1-(2,4,5-trimethoxyphenyl)-2-aminopropane
frrom Acorus calamus oil
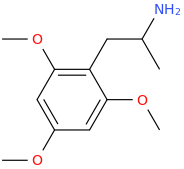
APPLE TREE AMPHETAMINE
1-(2,4,6-trimethoxyphenyl)-2-aminopropane
Time Was Ended Recently During The Age Of Aquarius, So Let The Sunshine In!
Last edited by a moderator:
Gaffy
Bluelighter
- Joined
- Oct 27, 2018
- Messages
- 1,210
Your images aren't showing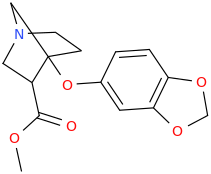
I'M WITH STOOPID
1-aza-3-carbomethoxy-4-(oxapiperonyl)norbornane
NITRODAPSONE
di(4-nitrophenyl)sulfone
broad spectrum antimicrobial for acne, Leprosy (Lou Hansen's disease), syphilis, pinta, yaws, Lyme's disease etc.
MIAGRA
1-methylpiperazin-4-yl 3,4-methylenedioxyphenyl sulfone
possible erection enhancer

CON Q PISS ANT
1-methylpiperazin-4-yl phenyl sulfone
possible aphrodisiac
"Be Like The Ant."--The Bible.
sekio
Bluelight Crew
- Joined
- Sep 14, 2009
- Messages
- 21,994
I fixed them. The issue was that you had unescaped space chracters in the URLs.
For instance,
[ІМG]htxps://opsin.ch.cam.ac.uk/opsin/ parabenzoquinone.png[/ІМG]
should be
[ІМG]htxps://opsin.ch.cam.ac.uk/opsin/parabenzoquinone.png[/IMG]
For instance,
[ІМG]htxps://opsin.ch.cam.ac.uk/opsin/ parabenzoquinone.png[/ІМG]
should be
[ІМG]htxps://opsin.ch.cam.ac.uk/opsin/parabenzoquinone.png[/IMG]
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,439
I fixed them. The issue was that you had unescaped space chracters in the URLs.
For instance,
[ІМG]htxps://opsin.ch.cam.ac.uk/opsin/ parabenzoquinone.png[/ІМG]
should be
[ІМG]htxps://opsin.ch.cam.ac.uk/opsin/parabenzoquinone.png[/IMG]
Dear sekio,
Could you explain that again? I didn't understand it the first time. "htxps"?
sekio
Bluelight Crew
- Joined
- Sep 14, 2009
- Messages
- 21,994
Ignore the htpxs. I just had to put that to prevent the forum software from making it into a broken a hyperlink.
I don't know how I can rephrase this too much differently... you see the difference between these two lines?
[ІМG]httрs://opsin.ch.cam.ac.uk/opsin/ parabenzoquinone.png[/ІМG]
[ІМG]httрs://opsin.ch.cam.ac.uk/opsin/parabenzoquinone.png[/ІМG]
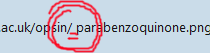
It's the space character you have in there. Whitespace (and some other special characters) is not valid in a URL and must be "escaped" by replacing it with a percentage sign and its ASCII code in hex. For the space character, the replacement is '%20'.
So, if you have a formula which needs a space character, every instance of it in the URL must be either removed, or replaced with "%20". I took the liberty of going through your recent posts and doing the needful.
I don't know how I can rephrase this too much differently... you see the difference between these two lines?
[ІМG]httрs://opsin.ch.cam.ac.uk/opsin/ parabenzoquinone.png[/ІМG]
[ІМG]httрs://opsin.ch.cam.ac.uk/opsin/parabenzoquinone.png[/ІМG]

It's the space character you have in there. Whitespace (and some other special characters) is not valid in a URL and must be "escaped" by replacing it with a percentage sign and its ASCII code in hex. For the space character, the replacement is '%20'.
So, if you have a formula which needs a space character, every instance of it in the URL must be either removed, or replaced with "%20". I took the liberty of going through your recent posts and doing the needful.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,439
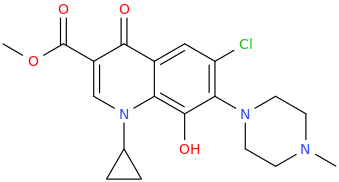
MARK GARNO SCHAEFER GARRETT QUARTERMAIN PHENNINGTON THE 3RD
8-hydroxy-3-carbomethoxy-1-cyclopropyl-6-chloro-4-oxo-7-(4-methylpiperazin-1-yl)quinoline
antibiotic
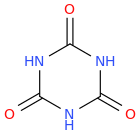
BARROW
1,3,5-triazinane-2,4,6-trione
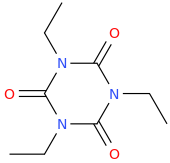
JENNIFER
1-ethyl-3-ethyl-5-ethyl-1,3,5-triazinane-2,4,6-trione
I AM A WRITER, A POET, A GENIUS, I KNOW IT.
Last edited: