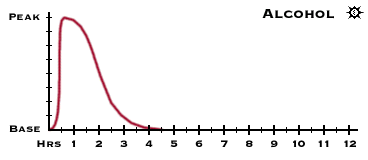
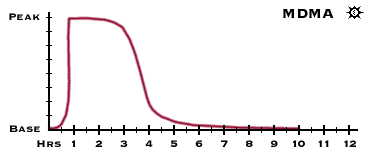
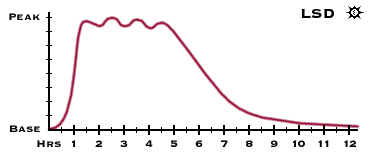
Alcohol a drug!! It's a natural body chemical isn't it?
Alcohol by any definition is a drug, but in some respects it differs from other rec drugs, as the body has evolved a particular mechanism to process it.
Simple alcohols (ethanol in particular) are natural body chemicals, produced in varying but small amounts during digestion and through normal metabolism. Occasionally, the amount of alcohol produced during digestion may be enough to cause mild intoxication, so the metabolic system is equipped to deal with relatively large amounts.
Moles, enzymes, 2 step & NAD+
On a molar basis, alcohol is consumed in far greater amounts than any other drug. A few drinks often involves more than a mole or two (1mole = 46g) being consumed. A “large” session may involve a MDMA molar equivalent of more than 1000 tablets, as 1 mole of MDMA HCl is 229g.
What this means is that the molar weight of any consumed alcohol requires an equimolar availability of enzymes and co-factors to facilitate metabolism. With long sessions, food or nutrients / vitamins are essential for the body to be able to regenerate cofactor NAD+.
Oxidation of 2 moles of alcohol requires ~ 1.5kg of cofactor NAD+
Following rapid absorption through the stomach, alcohol enters the liver via hepatic blood flow. The liver is the organ where all initial and most subsequent metabolism occurs; this being a 2 step oxidative process producing acetic acid which is then harmlessly eliminated.
In the liver, alcohol is first converted to acetaldehyde, approximately 75% by
alcohol dehydrogenase and ~25% by
mixed function oxidases
The second step of liver metabolism involves oxidizing ~75% of the toxic acetaldehyde to acetic acid via
aldehyde dehydrogenase . Around 25% is converted to acetic acid outside of the liver by cells employing
aldehyde oxidase
Both processes of alcohol metabolism involving
dehydrogenase enzymes also require cofactor
nicotinamide adenine dinucleotide in its protonated, hydride receiving form (NAD+). The availability of regenerated NAD+ limits the rate of alcohol metabolism and over 200 enzymes use NAD+ or NADP+ for normal body processes.
Alcohol competes with other NAD+ requiring substrates, and it is thought this may be involved in long term liver toxicity. Acetaldehyde needs to be metabolised quickly as it also is toxic to liver cells. Another area of potential liver toxicity from long term use is through rises in levels of fatty acid –ethanol esters which may also contribute towards a fatty liver which can lead to cirrhosis (fatty liver will show up on ultrasound, sometimes before it shows up in blood tests for liver enzyme GGT).
Getting rid of dangerous intermediates
As aldehydes are so toxic, when alcohol is consumed regularly - even in moderate amounts - the body increases levels of available
aldehyde dehydrogenase and
aldehyde oxidase . The majority of the former is in the liver, but the oxidase occurs throughout the body including the brain. These increases have been associated with observed variations in metabolite ratios from neurotransmitter catabolism, particularly serotonin. Altering enzyme ratios also has potential to adversely influence metabolism rates of natural endogens and alter metabolic routes for other sympathomimetic amines.
Inhibited Aversion
Drugs which competitively bind to
aldehyde dehydrogenase enzymes (e.g disulfiram) are sometimes used in aversion therapy during the treatment of alcoholism. On their own they have no noticeable physiological effect. However, when alcohol is consumed, these drugs prevent metabolism by this route and cause a build up of acetaldehyde in blood plasma. Plasma acetaldehyde produces an assortment of ghastly affects, including pain and nausea, but it is generally regarded as being harmless [?] It certainly discourages the user from wanting to drink while on the treatment.
Heart felt response to daily drinking
A good thing about daily, small amounts of alcohol (less than that required to cause CNS effects) is that the practice results in an elevation of plasma HDL, which in turn gives protection against coronary heart disease. Moderate drinking can give >50% protection in males. Pharmacology: Rang, Dale, Ritter 4th Ed.
Ethanol is the antidote for alcohol poisoning
Another interesting thing about ethanol is that it is the antidote for methanol poisoning!!
Ethanol has a higher affinity for binding with
alcohol dehydrogenase than does methanol. Ethanol is administered following methanol poisoning to prevent the formation of highly toxic formaldehyde which reacts readily with lipoproteins and ultimately destroys cells (the eyes are particularly sensitive to this). Methanol is then eliminated unchanged in the urine.
Engine suseptibility / fuel processing / head wear
In relation to the actions of the drug ethanol, I have really only mentioned the liver and briefly at that. Actions of ethanol on GABA, AMPA & NMDA receptors, it’s affect on Ca++ gated ion channels, ACTH –ADH, specific organ and general neuropharmacology are subjects in themselves. Alcoholics often obtain much of their calorie intake from alcohol which leads to withdrawal complications and a cold turkey period similar to that seen with heroin withdrawal. Mechanisms involved with many of these processes are also thought to contribute to general, nonreversible physical and mental decay seen with chronic alcoholism. It would appear the engine wear warnings for fuel containing too much ethanol equally applies to your body in relation to alcohol intake.
Spiritual respect
In defense of such a spiritually empowered chemical; when used sensibly and in moderation, the “spirit” can be a valuable ally - a medicine. It can facilitate introspection, temporarily relieve grief and reduce shyness. It can even improve health. And it does all this with a high margin for dose error.
Ethanol occurs everywhere in nature. From decomposing garbage to cellular function. It is always going to be with us, being an integral starting material for countless bio-chemicals. In the organic chemist’s lab, using simple chemistry, ethanol can produce an endless number of cyclic and bioactive compounds. In the alchemist’s world, the alcohol source, its refinement and subsequent purity are of the utmost importance. Ethanol truly is a piece of divine chemical Lego.
It’s just a pity general maturity regarding its use doesn’t reflect the length of time man has been harnessing its properties. Like most things, I guess you can’t have it all without a string or two attached