Limpet_Chicken
Bluelighter
- Joined
- Oct 13, 2005
- Messages
- 6,323
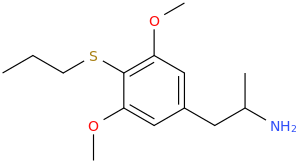
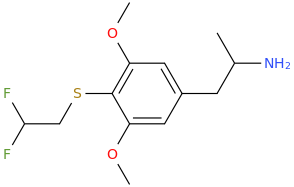
So, LC will soon be submitting 3-bromo-4-(1,1-difluoromethoxy)-5-methoxyamphetamine and the corresponding 3-bromo homolog of difluoromescaline, where bromine replaces the 3-position methoxy.
I did start a similar thread a while ago, but got few views and no feedback whatsoever. Surely somebody's going to bite on this. SOMEONE must know SOMETHING about mescaline/TMA/TMC (what I'd call the corresponding beta-ketones (if I try the cat version, it'll be in the form of the pthalimidopropiophenone, which should cleave in the stomachs acidic environment to liberate the cathinone, seeing how freakishly unstable primary beta-ketoamines can be from my experience trying, unsuccessfully to plug beta-C=O-2C-B, which one of the two attempted times actually turned into bright purple dimer on the journey from plastic pot with dry powder poured in, to adding 5ml H2O from a measuring syringe, drawing it up, it became inactive in the time between drawing up and journeying to chocolate starfish; the bk-2C-B itself was orally and intravenously (difficult in the extreme but doable if registering, pulling the syringe off the rig, holding the container with the beta-keto-2C-B close to the needle, left in the vein, then drawing up quickly and carefully reconnecting the rig to the needle. Not, in any case the most feasible mode of administration)
So, the pthalimidopropiophenone derivative, assuming there is enough aldehyde left for the cathinone, is a much more practical way to do things. Although it will of course make the time to onset and speed of comeup different from the unmodified amine, the time to onset being slower I'd imagine, and comeup being more prolonged due to gradual leaching of 3-bromo-4-(1,1-difluoromethoxy)-5-methoxycathinone into the system of the subject as the pthalimide group gets split off.
But first will be the phenethylamine or the TMA homolog (3-bromo-4-(1,1-difluoromethoxy)-5-methoxyphenisopropylamine.
It seems like 4-(1,1,1-trifluoromethyl) substitution pattern leads to extremely potent psychedelics in those phenethylamines and amphetamines where the rest of the substitution pattern of the aromatic ring corresponds to known active compounds (e.g DOTFM and 2C-TFM) and trifluoromescaline (where the para-methoxy is replaced by a difluoromethyl ether) is known to be active, IIRC at some 15-35mg or so, 60mg is reported as 'strong overdose' (https://en.wikipedia.org/wiki/Trifluoromescaline
Difluoroethoxy seems to work in the case of the difluoroethoxy homolog of 3C-E, potency is increased a little in terms of dose range being lower, and whilst its a little ambiguous (3C-E is stated to last some 8-12 hours whilst the difluoroethoxy homolog is reported as 10h) as I expected the duration sounds like its a little increased, presumably due to the fact that fluorine acts as a bioisostere of a sort of hydrogen and can only generally be removed in-vivo by means of lopping off the fluorinated part wholesale, at carbon or heteroatom bonds linking the fluorinated portion of the molecule, leaving a fluorinated fragment, or occasionally as fluoride ion, in such compounds as halothane), but usually, when fluorine is attached to carbon at least, it isn't coming off. Although this is of more importance in fluoroalkyl groups and fluoroaryls, cleavage at the ether bridge of course makes it more practical.
The ONLY instance of an electron-withdrawing substitution, a halide or otherwise, is 2-bromomescaline, which is listed in wikipedia as having a quite significantly greater Ki value by an order of magnitude, and 2-bromo-4,5-MDA which is less potent than MDA (dose listed as some 300mg)
https://en.wikipedia.org/wiki/2-Bromomescaline 513nm and 215nm at 5HT2a and 5HT1a respectively)
Nothing with a halogen, cyano, nitro etc at the 3'carbon however.
So guys, take this one apart, and throw me a bone maybe?
I did start a similar thread a while ago, but got few views and no feedback whatsoever. Surely somebody's going to bite on this. SOMEONE must know SOMETHING about mescaline/TMA/TMC (what I'd call the corresponding beta-ketones (if I try the cat version, it'll be in the form of the pthalimidopropiophenone, which should cleave in the stomachs acidic environment to liberate the cathinone, seeing how freakishly unstable primary beta-ketoamines can be from my experience trying, unsuccessfully to plug beta-C=O-2C-B, which one of the two attempted times actually turned into bright purple dimer on the journey from plastic pot with dry powder poured in, to adding 5ml H2O from a measuring syringe, drawing it up, it became inactive in the time between drawing up and journeying to chocolate starfish; the bk-2C-B itself was orally and intravenously (difficult in the extreme but doable if registering, pulling the syringe off the rig, holding the container with the beta-keto-2C-B close to the needle, left in the vein, then drawing up quickly and carefully reconnecting the rig to the needle. Not, in any case the most feasible mode of administration)
So, the pthalimidopropiophenone derivative, assuming there is enough aldehyde left for the cathinone, is a much more practical way to do things. Although it will of course make the time to onset and speed of comeup different from the unmodified amine, the time to onset being slower I'd imagine, and comeup being more prolonged due to gradual leaching of 3-bromo-4-(1,1-difluoromethoxy)-5-methoxycathinone into the system of the subject as the pthalimide group gets split off.
But first will be the phenethylamine or the TMA homolog (3-bromo-4-(1,1-difluoromethoxy)-5-methoxyphenisopropylamine.
It seems like 4-(1,1,1-trifluoromethyl) substitution pattern leads to extremely potent psychedelics in those phenethylamines and amphetamines where the rest of the substitution pattern of the aromatic ring corresponds to known active compounds (e.g DOTFM and 2C-TFM) and trifluoromescaline (where the para-methoxy is replaced by a difluoromethyl ether) is known to be active, IIRC at some 15-35mg or so, 60mg is reported as 'strong overdose' (https://en.wikipedia.org/wiki/Trifluoromescaline
Difluoroethoxy seems to work in the case of the difluoroethoxy homolog of 3C-E, potency is increased a little in terms of dose range being lower, and whilst its a little ambiguous (3C-E is stated to last some 8-12 hours whilst the difluoroethoxy homolog is reported as 10h) as I expected the duration sounds like its a little increased, presumably due to the fact that fluorine acts as a bioisostere of a sort of hydrogen and can only generally be removed in-vivo by means of lopping off the fluorinated part wholesale, at carbon or heteroatom bonds linking the fluorinated portion of the molecule, leaving a fluorinated fragment, or occasionally as fluoride ion, in such compounds as halothane), but usually, when fluorine is attached to carbon at least, it isn't coming off. Although this is of more importance in fluoroalkyl groups and fluoroaryls, cleavage at the ether bridge of course makes it more practical.
The ONLY instance of an electron-withdrawing substitution, a halide or otherwise, is 2-bromomescaline, which is listed in wikipedia as having a quite significantly greater Ki value by an order of magnitude, and 2-bromo-4,5-MDA which is less potent than MDA (dose listed as some 300mg)
https://en.wikipedia.org/wiki/2-Bromomescaline 513nm and 215nm at 5HT2a and 5HT1a respectively)
Nothing with a halogen, cyano, nitro etc at the 3'carbon however.
So guys, take this one apart, and throw me a bone maybe?