-
Select Your Topic Then Scroll Down
Alcohol Bupe Benzos Cocaine Heroin Opioids RCs Stimulants Misc Harm Reduction All Topics Gabapentinoids Tired of your habit? Struggling to cope?
Want to regain control or get sober?
Visit our Recovery Support Forums -
OD Moderators: Keif’ Richards | negrogesic
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
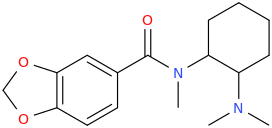
RCs Novel opioid 3,4-Methylenedioxy-U-47700
- Thread starter roi
- Start date
Wiserthanearlier
Bluelighter
- Joined
- Feb 6, 2017
- Messages
- 676
Any notes on expected activity or differences from u-47700?
It's mdu4700 lol.... Really though what's the difference with the change? Any significance in it sharing similarity with mdma? I'm not expecting it to be like mdma, but maybe it might have less shitty side effects? Or more? S
Who knows I'm interested to see results from the first Guinea human
Who knows I'm interested to see results from the first Guinea human
I never assumed it was like mdma just shared the 3'4 methyldioxy instead of the dichloro as you said..... Maybe there's a dichloro analog of mdma {dcma?) and the increase or decrease in effect or lack of change would be something similar if such an mdma analog exists... Of course it's more complicated than that, but it just leaves me curious how that makes it different.... The vendor claims similar potency, but they also do about the notoriously ineffective and toxic u48800 and/or u49900.
In surprised you would think I'm that ignorant to think it would be similar in affect or anything to mdma vs just having a similar partial structure to an already well known compound with many analogs used in place yet still raining as the best except mda to some... I think using DCA and MDA as a comparison will make my point.
https://en.m.wikipedia.org/wiki/3,4-Dichloroamphetamine
https://en.m.wikipedia.org/wiki/3,4-Methylenedioxyamphetamine
A good base compound can either be a magic compound or a neurotoxic compound by the simple difference U47700 and thus compound shares. Now that doesn't mean this new compound is safe in fact it can be more dangerous? Although if it does share the quality traits of the above compounds then wouldn't it be logical to assume this is likely an improvement? Like I said no one should assume it's safe until there are some reports of someone smart enough to do an allergy test then start low, but I can't help wonder if this might be a suburbanites) significant change or not.
In surprised you would think I'm that ignorant to think it would be similar in affect or anything to mdma vs just having a similar partial structure to an already well known compound with many analogs used in place yet still raining as the best except mda to some... I think using DCA and MDA as a comparison will make my point.
https://en.m.wikipedia.org/wiki/3,4-Dichloroamphetamine
https://en.m.wikipedia.org/wiki/3,4-Methylenedioxyamphetamine
A good base compound can either be a magic compound or a neurotoxic compound by the simple difference U47700 and thus compound shares. Now that doesn't mean this new compound is safe in fact it can be more dangerous? Although if it does share the quality traits of the above compounds then wouldn't it be logical to assume this is likely an improvement? Like I said no one should assume it's safe until there are some reports of someone smart enough to do an allergy test then start low, but I can't help wonder if this might be a suburbanites) significant change or not.
swim_instructor
Greenlighter
- Joined
- May 30, 2017
- Messages
- 5
Got quite a large sample of this this morning. Threshold seems to be around the 60mg range sniffed. No oloiod tolerance at the moment, however it takes about 60mg morphine or 40mg oxy for me to feel anything euphoric. U-47700 would work wonders for me around 20-40 mg so far this analogue is not anything close to the original in my opinion. I Will try other ROI as time goes on. So far only a tease at 60mg insuffilated.
busted.produk
Bluelighter
Got quite a large sample of this this morning. Threshold seems to be around the 60mg range sniffed. No oloiod tolerance at the moment, however it takes about 60mg morphine or 40mg oxy for me to feel anything euphoric. U-47700 would work wonders for me around 20-40 mg so far this analogue is not anything close to the original in my opinion. I Will try other ROI as time goes on. So far only a tease at 60mg insuffilated.
This is directed towards the thread(not at you swim_instructor), im just using the quote function as reference.
Dont forget to titrate new compounds folks.
For harm reduction purposes i want to emphasize that just because the 1st review(and 1st post from new member) of this compound states 60mg is a "tease", you should still treat it with caution.
This should go without saying but i felt i should add this because ive read a lot of posts lately that seem a little careless in the department of experimentation.
Disrespect or discreditation are neither my intent, swim_instructor.
Just want folks to be safe while having fun....
Zoops
Greenlighter
U-47700 is shite. This is shite too. Too much kappa agonist activity for my taste. It's crazy-making stuff. Fentanyls are a MUCH better class of opioid to research.
Did you even try this stuff? The way you posted was extremely vague and harmful suggesting people use fentanyl.U-47700 is shite. This is shite too. Too much kappa agonist activity for my taste. It's crazy-making stuff. Fentanyls are a MUCH better class of opioid to research.
I'll also add it is definitely important to allergy test a single mg of any compound at most before a real dose, which should also start low no more than 5 mg then 10, 20, 30, 40 etc.
swim_instructor
Greenlighter
- Joined
- May 30, 2017
- Messages
- 5
Thank you busted. Yes I started very low (2mg and eventually got to 60) first post quite frankly because I have not posted in possibly 10 years and I don't think I have my original email or username. Just did not see any reports on this compound so I thought I would quickly jot down my first impression. Thank you busted, and thank you community for clarifying in regards to titration. Compared to the original this compound is not worth the money.from now on I will remember that may people need very accurate step by step, and not assume that if you are digging through an obscure thread like this you know risks involved. I did love this form for the HR. Oh I have the .HCL vaping is proving to be much more pleasurable. (Last edit. Feel free to ask for clarity if needed)
Last edited:
LaGaFàNi
Bluelighter
- Joined
- Jun 28, 2016
- Messages
- 93
It's quite normal for this to be needing a higher dose than the original to accomplish anything, as the benzodioxol ring has to be opened by metabolism for it to ressemble U-47700 binding-wise.
I think the active form of this (if you overlay structure with morphine) would be the 3,4-(DiHydroxy-Deschloro)-U47700; 3-Ethoxy-4-Hydroxy-DesCl-U-47700 would ressemble EthylMorphine and 3-Hydroxy-4-Ethoxy has it ressemble Morphine (when overlayed). So the closed ring structure allows it to better pass the BBB, but should be less active than it's metabolites as it doesn't ressemble morphine's structure.
Another reason for it to be less potent is that a part of the dose might first be metabolized by N-DiMethylation which renders it inactive before ring-opening.
Just as for codeine and it's derivatives, things like DPH or white grapefruit should potentiate enzymes catalyzing the ring-opening making it more potent (I suppose), and co-administration with BBB passing anti-oxidants should also potentiate it/ make it last longer. (All pure speculation ofc)
I think the active form of this (if you overlay structure with morphine) would be the 3,4-(DiHydroxy-Deschloro)-U47700; 3-Ethoxy-4-Hydroxy-DesCl-U-47700 would ressemble EthylMorphine and 3-Hydroxy-4-Ethoxy has it ressemble Morphine (when overlayed). So the closed ring structure allows it to better pass the BBB, but should be less active than it's metabolites as it doesn't ressemble morphine's structure.
Another reason for it to be less potent is that a part of the dose might first be metabolized by N-DiMethylation which renders it inactive before ring-opening.
Just as for codeine and it's derivatives, things like DPH or white grapefruit should potentiate enzymes catalyzing the ring-opening making it more potent (I suppose), and co-administration with BBB passing anti-oxidants should also potentiate it/ make it last longer. (All pure speculation ofc)
swim_instructor
Greenlighter
- Joined
- May 30, 2017
- Messages
- 5
Interesting Laga . Today I will load up on grapefruit, not holding my breath though.... Very reminiscent of a buprenorphine buzz.... I have not let it ride it's full length. Going to find out how long it lasts.
Last edited:
roi
Bluelighter
- Joined
- Sep 2, 2013
- Messages
- 1,545
U-47700 is shite. This is shite too. Too much kappa agonist activity for my taste. It's crazy-making stuff. Fentanyls are a MUCH better class of opioid to research.
The U-47700 µ to κ ratio is 200:1, you won't notice that lol. It's probably the lack of δ-opioid receptor activity that makes it different compared to other opioids.
swim_instructor
Greenlighter
- Joined
- May 30, 2017
- Messages
- 5
OK. So my conclusion is that this chemical is crazy addictive with out much euphoria. I was well over 300mg a dose, and just using to use. (5g in 3 days) now now WD are worse than dope, bupe, oxy. This is a chemical where the risk is not worth the reward... 0 tolerance to worst WD ever in three days. = not fun sorry for being so short with reports. PASS ON THIS CHEM.