-
N&PD Moderators: Skorpio | thegreenhand
-
Neuroscience & Pharmacology Discussion Welcome Guest
Posting Rules Bluelight Rules Recent Journal Articles Chemistry Mega-Thread FREE Chemistry Databases! Self-Education Guide
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Simplest molecule filling morphine rule?
- Thread starter dread
- Start date
No matter how often I study it, nomenclature is never obvious to me. It appears that the most basic morphine rule filling molecule isn't ENTIRELY bereft of applications. It's a substructure for an antidepressant known as nafenadone. Not exactly what I'd HOPED out of the molecule, but edifying, nonetheless.
Has anyone taken a good hard look at the milnacipran (Savella) structure? By itself, it doesn't fit the morphine rule. But if someone decided to methylate the primary amine to a tertiary amine, all morphine rule constraints are satisfied. If the carbonyl group survives the di-amination reaction, the end result can be found at PubChem or ChemSpider, but precious little is said of its pharmacodynamics. Thoughts, anyone?
BTW, the reason you're not seeing a structural formula for both molecules is that I don't know how to put them on here.
BTW, the reason you're not seeing a structural formula for both molecules is that I don't know how to put them on here.
Hammilton
Bluelighter
- Joined
- Sep 2, 2008
- Messages
- 3,435
omg wow two phenethylamine-derived drugs stuck together. holy jesus what an idea....
Perhaps giving a little background to your posts could be a little more useful...
1. It doesn't fit the morphine rule, so why is it in this thread?
and
2. What does it have to do with anything?
Perhaps giving a little background to your posts could be a little more useful...
1. It doesn't fit the morphine rule, so why is it in this thread?
and
2. What does it have to do with anything?
dread
Bluelighter
- Joined
- Aug 20, 2008
- Messages
- 2,974
Shit, I see I posted the wrong image... sec.
That's what you get for posting while sleep deprived. And having lots of random chemical doodles on your HD, I guess...
Now there is the one I meant to post here. Now, can anyone think of any simpler molecule fitting the rule?
Of course I do realize this molecule probably wouldn't be very stable at all... lots of strain. But I can't think of any way to make a molecule that fits the morphine rule any simpler... that is, one with the least atoms, counting hydrogens also.
That's what you get for posting while sleep deprived. And having lots of random chemical doodles on your HD, I guess...
Now there is the one I meant to post here. Now, can anyone think of any simpler molecule fitting the rule?
Of course I do realize this molecule probably wouldn't be very stable at all... lots of strain. But I can't think of any way to make a molecule that fits the morphine rule any simpler... that is, one with the least atoms, counting hydrogens also.
Last edited:
Back to 4-phenylquinuclidine. I think it's lighter than LS-47438 by 2 hydrogens, but you end up with a polycyclic molecule, with a rather nasty therapeutic index. I think its pharmacodynamics are somewhat comparable to a third-rate nerve gas.
DREAD, is there a structure-activity rule or trend that suggests that the molecule I described might be an antagonist? I know that converting the codeine or morphine molecules to a ketone increases the per-milligram potency, as does converting the keto-analog to a hydrogenated or oxygenated analog. Does the presence of a cyclopropane group suggest partial agonist or antagonist properties?
Hey HAMMILTON, I saw your quip about the "two phenethylamines stuck together" and requesting "background on your posts" as well as the query "what does it have to do with anything". Should I assume that this was directed at me and that you're STILL in the dark about it?
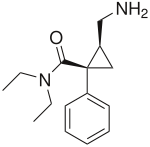
The structure depicted is NOT what I had in mind, but I can't get structures to post here. The SMILES depiction of what I was talking about with the primary amine substituted by a tertiary amine or a piperdine is as follows, respectively:
dimethylamine: C1C(C1N(C)C)(C(=O)N(CC)CC)C2=CC=CC=C2
piperidine: C1C(C1N2CCCCC2)(C(=O)N(CC)CC)C3=CC=CC=C3
I hope that this clarifies the manner in which I was "wondering aloud". I'm not a pharmacologist, and I don't even play one on TV. I'm just barely a chemist, but the mystery of structure-activity relationships is the source of considerable fascination for me. If I wandered(OR wondered) too far afield from the topic of this string for the comfort of ANYONE, then I apologize unreservedly.
BTW, thanks DREAD for the input about the cyclopropylmethyl group confering antagonist/partial agonist activity when attached to the amine on phenanthrene based opioids. I wonder if this generalizes to the phenyl-piperidines as well, without making the chemical prone to producing seizures, psychosis, or other intolerable side effects.
The structure depicted is NOT what I had in mind, but I can't get structures to post here. The SMILES depiction of what I was talking about with the primary amine substituted by a tertiary amine or a piperdine is as follows, respectively:
dimethylamine: C1C(C1N(C)C)(C(=O)N(CC)CC)C2=CC=CC=C2
piperidine: C1C(C1N2CCCCC2)(C(=O)N(CC)CC)C3=CC=CC=C3
I hope that this clarifies the manner in which I was "wondering aloud". I'm not a pharmacologist, and I don't even play one on TV. I'm just barely a chemist, but the mystery of structure-activity relationships is the source of considerable fascination for me. If I wandered(OR wondered) too far afield from the topic of this string for the comfort of ANYONE, then I apologize unreservedly.
BTW, thanks DREAD for the input about the cyclopropylmethyl group confering antagonist/partial agonist activity when attached to the amine on phenanthrene based opioids. I wonder if this generalizes to the phenyl-piperidines as well, without making the chemical prone to producing seizures, psychosis, or other intolerable side effects.
sarbanes
Bluelighter
- Joined
- Mar 8, 2007
- Messages
- 481
Hey HAMMILTON, I saw your quip about the "two phenethylamines stuck together" and requesting "background on your posts" as well as the query "what does it have to do with anything". Should I assume that this was directed at me and that you're STILL in the dark about it?
The structure depicted is NOT what I had in mind, but I can't get structures to post here. The SMILES depiction of what I was talking about with the primary amine substituted by a tertiary amine or a piperdine is as follows, respectively:
dimethylamine: C1C(C1N(C)C)(C(=O)N(CC)CC)C2=CC=CC=C2
piperidine: C1C(C1N2CCCCC2)(C(=O)N(CC)CC)C3=CC=CC=C3
I hope that this clarifies the manner in which I was "wondering aloud". I'm not a pharmacologist, and I don't even play one on TV. I'm just barely a chemist, but the mystery of structure-activity relationships is the source of considerable fascination for me. If I wandered(OR wondered) too far afield from the topic of this string for the comfort of ANYONE, then I apologize unreservedly.
BTW, thanks DREAD for the input about the cyclopropylmethyl group confering antagonist/partial agonist activity when attached to the amine on phenanthrene based opioids. I wonder if this generalizes to the phenyl-piperidines as well, without making the chemical prone to producing seizures, psychosis, or other intolerable side effects.
No, it does not generalize to meperidine and the like. This is one of the reasons why, although morphine shares certain structural characteristics with meperidine (similar phenyl-pperidine structure, alkylation of the amine in morphine with cyclopropyl or allyl leads to antagonist, but not wit the demerol type), the agonist target mechanisms between morphine type and 4-phenylpiperidines, is proposed to be different.
I believe, generally speaking, cyclopropylmethyl on the amine for morphine (morphinan) skeleton ALWAYS leads to antagonist. More highly substituted oripavines and metopons are the exception (buprenorphine falls into 1st category), but bup isn't a morphine derivative...it's a thebaine derivative.
dread
Bluelighter
- Joined
- Aug 20, 2008
- Messages
- 2,974
cyclopropylmethyl on the amine for morphine (morphinan) skeleton ALWAYS leads to antagonist.
Not so. N-cyclopropylmethyl combined with 14-phenylpropoxy gives a potent agonist.
The 14-phenylpropoxy substitution appears to confer potent μ-opioid agonist activity, even when combined with substitutions such as N-cyclopropyl or N-allyl, which normally result in μ-opioid antagonist compounds.
http://en.wikipedia.org/wiki/14-Phenylpropoxymetopon
Last edited: