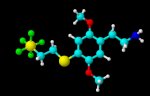
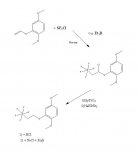
given that sulfur tetrafluoride is a good fluorinating agent don't expect an aryl tetravalent sulfur fluoride to be anything less than lethal.
the hexavalent sulfur compound might be less dangerous and more stable. But pretty much sulfur hexafluoride is the only inert and therefore non toxic sulfur fluoride.
the hexavalent sulfur compound might be less dangerous and more stable. But pretty much sulfur hexafluoride is the only inert and therefore non toxic sulfur fluoride.