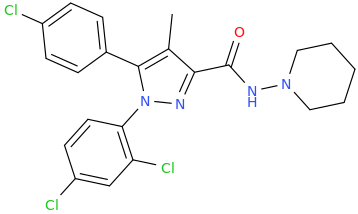
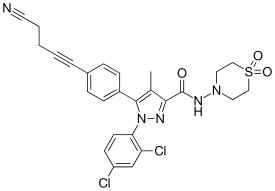
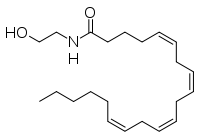
Why does diastereomeric recrystallisation work?
What are the molecular reasons behind why 2 enantiomers can't be separated with chromotography but 2 diastereomers can?
It boils down to solvation: the solvation shell around a diastereoisomer pairs is different for each of the diastereoisomer. So their physical properties including their Rf is different. And also because of that, they crystallize under different conditions hence they can be separated by selective fractional recrystallization.
On the other hand, solvation of an enantiomeric pair is the same for both enantiomers. So their physical properties will be the same in solution (ie for Rf: the eluting solvent and the stationary phase can't tell the difference so to speak). UNLESS the solvent used is chiral in that case the solvation will give 2 ( L(+)S(+) and L(-)S(+) not 1 solvated entities (L(+)-S. Kind of like the non-covalent (VanderWaals only) complex equivalent of diastereoisomers! chiral compound solvated by chiral solvent or complexed by chiral chromatographic media. Hence the use of chiral stationary phase to separate enantiomers... does it make sense??
Last edited: