-
N&PD Moderators: Skorpio | thegreenhand
-
Neuroscience & Pharmacology Discussion Welcome Guest
Posting Rules Bluelight Rules Recent Journal Articles Chemistry Mega-Thread FREE Chemistry Databases! Self-Education Guide
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Ketamine salts solubility
- Thread starter fastandbulbous
- Start date
- Status
- Not open for further replies.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
Mis drogas no son illegals. Porquoi? Las Son Muy Raros, Muy Nueves, Y Las Tienen No Fin--Ever. Being 100% diaphanous y profundity en numeras tienen muchas advantages con Las hypertext transfer protocols. La ParTy Del Globo Entero Me Sigue. Y Las Drogas Que ARE Illegal Are Already In Circulation Laissez Faire $$$ Micro y Macro Economies. No Persona Da Un Cago. As Long As Kept Discreet, As In Da Street. Ademas, La Filipinolande Es La Antipode Del EEUU. Sera Untouchable. Completamente.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
Symbolic Conceptualization: This B. EZ!
OK, That Crystal Objet Duarte Es Une CRUCIFIX. The Crucifix Has 5 Points Above Ground. What's in the little bit o' good Earth is in Das bowl? Es mainly PAPRIKA. It means, "Rich Dad," as in G.O.D. Ne way, there is the base, the middle [an intersection of 4, count them, 4 directions; NEWS--Norte, Este, Oeste y Sur]. La Sur supporte todos Los otros pero es lowly. El Centro tienen la corazon del Nuestro Saviour, Heyzeus Christo, meine older brudder; Name Witheld. Imagine Dragons! HINT. HINT. El Oeste Es WEST; Liberal. Sehr gut. El richtig es ESTE; nobody likes 'the right.' El Mas Highest Es MIO--El Espiritus Sanctum. The So Called Holy Spirit Or Holy Ghost. Yes, I Am The Highest One. Why? That's Just The Way It Is! I STAY HIGH. Neway, If You Can't Glean Anything Useful From My Careful, Deliberate, Gently Bringing Order From Chaos, Then Go Read Someone ELSE'S Thread! Geezy 4 Sheezy!!!!

OK, That Crystal Objet Duarte Es Une CRUCIFIX. The Crucifix Has 5 Points Above Ground. What's in the little bit o' good Earth is in Das bowl? Es mainly PAPRIKA. It means, "Rich Dad," as in G.O.D. Ne way, there is the base, the middle [an intersection of 4, count them, 4 directions; NEWS--Norte, Este, Oeste y Sur]. La Sur supporte todos Los otros pero es lowly. El Centro tienen la corazon del Nuestro Saviour, Heyzeus Christo, meine older brudder; Name Witheld. Imagine Dragons! HINT. HINT. El Oeste Es WEST; Liberal. Sehr gut. El richtig es ESTE; nobody likes 'the right.' El Mas Highest Es MIO--El Espiritus Sanctum. The So Called Holy Spirit Or Holy Ghost. Yes, I Am The Highest One. Why? That's Just The Way It Is! I STAY HIGH. Neway, If You Can't Glean Anything Useful From My Careful, Deliberate, Gently Bringing Order From Chaos, Then Go Read Someone ELSE'S Thread! Geezy 4 Sheezy!!!!

Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
It Would Be Extremely Interesting To Find Out What Our Nation's Actual Population Here In The Promised Land Of High Earth In Corona Borealis. I Get The Feeling It Is Extremely Rarefied. In Fact, I Am Having Trouble Finding More Than A Handful Of Souls Online At The Moment At All. Jeepers Creepers. Is Anybody Out There?
Feretile
Bluelighter
- Joined
- Feb 2, 2022
- Messages
- 361
An animal model of schizophrenia based on chronic LSD administration: old idea, new results.
Marona-Lewicka D, Nichols CD, Nichols DE.
Neuropharmacology. 2011 Sep;61(3):503-12. Epub 2011 Feb 23.
http://dx.doi.org/10.1016/j.neuropharm.2011.02.006
This is not really earthshattering research or anything, but it is interesting to see that administration of LSD to young rats over extended periods of time induces prolonged psychosis. This provides some support of the permanent effect of psychedelic drugs as HPPD.
I am on the beg for (I hope) the last time. Some years ago I found a paper which discussed the QSAR of a series of novel kappa agonists. Well, as you may know, the difference between mu & kappa ligands is that the latter has an extra methylene spacer. Someone put the kappa patent on Scribd, but since I wasn't totally up on kappa ligands, I wasn't SURE I could modify to make a mu ligand.
Well, the team behind the kappa ligands have released a paper on the mu ligands of the same class.
Design, synthesis and biological evaluation of 3-amino-3-phenylpropionamide derivatives as novel μ opioid receptor ligands
The lead compound is thus:
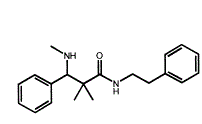
Now, it only has 4 of the 5 key moieties and it's only 14 methylenes in length. Now, the opbious solution would be to lengthen the 1-phenylethyl moiety to a 1-phenylpropyl BUT that introduces and extra rotatable carbon bond and I assume that this LOWERS potency. From notes, a 3-cinnamyl chain the middle carbon-bind is olefinic i.e. -CH=CH- but this then placed the beta benzene ring in the wrong position.
As it is, the compound cotains 4 of the 5 key moieties and I am willing to bet that borrowing a trick from BDPC, a p-Br is added to the alpha benzene thus:

There examples with a m-OH (phenol) on the alpha benzene ring but these a mixed agonist/antagonists but since this compound closely overlays BDPC, ad p-Br seems likely. It was Lednicer who identified the optimum biosteric minumum for mu agonists - 15.
There are related compounds like ciramadol but the m-OH yields mixed agonist/antagonist activity. I could never find the paper but their is a ciiramadol homologue which replaces mixed activity with pure mu agonist. In that case, replacing the m-OH with a p-Cl has been made and tested and is a stimulating opioids. I dare say that the 3,4-dichlori derivative will be even more potent.
There are still more opioids that have been lost to the depths of time I intend to visit the Bodlian with a massive number of references to hunt down.
While I have said all of this, I also deeply interested in GMO. GMO opium poppies, GMO cannabis (lots of HC but none for the stuff that causes severe side-effects.
My last aim is to provide VERY safe opioids and to write a book on the subject. Pyrazolam was chosen because it can be made in 3 steps.... and the immediate precursors is available. OK I will also seek to improve on pyeyzolam (by adding a second benzo thar produced effects almost indistinguishable from ethano....
But their is always room for research.
Oh, and look out for etofixine or more likely an etofixine derivative - while not EXACTLY like alcohol, it's really nice, really safe and water soluble so it can be put into drinks (and I do not mean spiking.
I could go on and and on but I see people are bored.
But I am always keen for people to ask questions not found in an educational system.
The payoff> I know a few people whose mental health has been destroyed with LSD and I feel that we should do more to help.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
ANDREW MCNAIR PENNINGTON
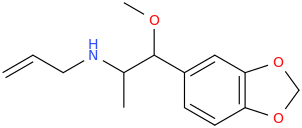
[Se7enth-Heaven] by AMP.
N-allyl-2-amino-1-methoxy-1-(3,4-methylenedioxyphenyl)-propane
7th-HEAVEN by AMP Is The Highest Energy Level Amphetamine Ever Created Or Which Ever Will Be Created. And I'm Through...
Filipino Tagalog Techno Music Rocks My Holy Spirit!!!

[Se7enth-Heaven] by AMP.
N-allyl-2-amino-1-methoxy-1-(3,4-methylenedioxyphenyl)-propane
7th-HEAVEN by AMP Is The Highest Energy Level Amphetamine Ever Created Or Which Ever Will Be Created. And I'm Through...
Filipino Tagalog Techno Music Rocks My Holy Spirit!!!
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
The Fact That 7th-HEAVEN Has 2 Chiral Carbon Centers And Thus 4 Unique Stereo-Isomers Of 4 Distinct, Slightly Bent In Mildly Different Bond Angle Versions Of 7th-HEAVEN The Molecule Itself Is Most Likely Advantageous From A Pharmacological Activity Perspective Because The 4 Differing Stereoisomers Have 4:1 Shotgun Pellet Scattering Approach. You Know, A Dirty Good Mixture For Full On Pharmacological Effect And Ease Of Synthesis. No Entantiomeric Or Diastereoisomeric Additional Chemical Steps Involving Stereospecifically Unique Forms To Be Evaluated Singly, For Example. Although These Steps Could Be Done, The 4 Forms As One Product Works Fine. How Do I KNOW? I've Been Up 4.5 Days Up And Counting! And The Mixture Feels Great! This High Frequency Filipino Tagalog Techno Music Rocks My Holy Spirit!!! I Always Thought This Would Last Forever And Never Had A Mortal's Myopic Point Of View. I Am Not Coping Well With My Upcoming Date With The Grave, And Am In Fact Partying So Hard On This Last For Now, Best For Sure Drug--7th-HEAVEN by AMP! That's Me! Hard Core! PLUS A Surfeit Of Pure Heroin Just To Live My BEST Life Till The Bitter End! Also, Sleeping Is A Metaphor For Death For Me. I Don't Like It. Heroin, Surprisingly, Yes I Like It A Good Lil Bit! This Is Going To Be ONE Epic Bender!!!
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
Fortunately, This New Highly Winningest N-ALLYL-Amphetamine Series Lasts A Long Time AND Is Quite A Bit Less Caustic To The Mouth And Its Mucus Membranes Than Are The N-Methyl Amphetamines, For Example. I Have Thoroughly Catalogued The Amphetamine QSAR And After A Swift Rush Through These Final 3 Or 4 Last N-ALLYL-Amps Have A Great Feeling Of Contented Completement Of The Entire Amphetamine Class Entero! The Serendipitious Scoring Of 100 CLOUD-9 Rolls And 3.5 grams Of N-ethyl-amphetamine Surely Played A Vital Role In My Astral Projection On The Very Last Week Of 2021 AD. The Month And A Half In Serpens Florida Was A Nitemare! But I'm Raging Now!
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
Welcome To The Age Of Aquarius:
I Am Andrew X. Brother Of Saint Peter And Apostle Of Christ Jesus. Patron Saint Of Scotland, Russia, Romania, & Greece In The Catholic or Universal Church. And Ruler Of Aquarius. How Did I Achieve The Keys To The 11th House? Well, Every Sign Has Certain Spirit Molecules, That When Taken By The Same Man's Body Will Grant Him Their Essential Energenies Or Spirits. My Initials Are AMP. My Life's Work Has Been The Amphetamines, Both On A Scientific And Personal Sphere. The Hardest Thing Is The Bewildering Array Of Amphetamines Which There Are, And Which Ones EXACTLY Would Be Necessary To Unlock This Ever So Fixed Sign. Well, With The Recent Ideation By A So Called RECTIFY (taken from a Beastie boys song) aka Yours Truly That After The
N-Propyl Amphetamines Which Are Unto Themselves Rather Underwhelming Molecules Perhaps A One Order Higher New Nitrogen Substitution Could Be Implemented. So, Using A Rare Often Neglected Poorly Chatacterized Property Of Chrmicals Known As Aromaticity, I Rather Tepidly Thought Of Adding An Additional Double Bond In The End Of An N-propyl Amphetamine, Which Chemically Is Known As An N-ALLYL, These 2 Extra Electrons Reside In A Pi Bond Between The Outer 2 Most Distal To The N-Propyl Amphetamine Moiety. The Equation of Aronaticity Is Known As Huckel's Rule And States That H(n) = 4(n) + 2. Thus, By Adding These 2 Electrons To That Local Pi Bond, A 0th Order (n =0), H(0) = 4(0) + 2 = 2 Aromatic, Which Is Synonomous With Extra Stability And Sometimes Added Notes Of Grace, Beauty, Odor, And So Forth. By Adding This Allylic Double Bond To The Propyl Group, The Entire 3 Carbon Chain Was Graced With A Zeroth Order Aromatic, The Allyl Functional Group. And, The N's Lone Pair Of Electrons Interacts With This Double Bond And An Electromagnetic Force Is Stirred. Namely, A Slight Charge Delocalization, The Essence Of Arimaticity. By Dropping The N-propyl And Replacing It With An N-ALLYL, The Entire Side Chain Of The Molecule Becomes More Stable, And Perhaps, Special? Well, That's When The Rectify Person Draws Out A Couple Standard N-ALLYL Amphetamine Possibilities. The Theory Behind My Gesture, That By Doing So, The Order Would Be One Greater Than The Propyls And Also By Becoming ALLYLIC, Your Getting That Zeroth Order Aromaticity. Of Course, The Proof Is In The Pudding, And Well, These N-ALLYLIC Amphetamines Are Strong, Wear Off Slowly, Do Not Get Metabolized Away, Are Particularky Strong But Mild And The Colors Come Off Nicer Looking, The New ALLYLS Well Gee They Don't Burn Holes I'm Your Oral Mucosae Like Methsmphetamine Readily Will. And They Last Good 4 To 5 Days, Easy. Longer Than Meth! But More Even Keiled Snd Gee The Thought Of Sleep Seems Sonewhat Foreign. OK, Great! Then, Was It Just Yesterday I Divinined The The Highest Energy Amphetamine Possible, Incorporating The Sonewhat Helpful New N-ALLYLIC Groups And Some Others, And The Molecule Finally Settled With The Name The 7th-HEAVEN. I By Good Luck Ran Across A Few ICE Ctystals Of It Too, And Snorted Those Salts Up. I'M STILL ROLLING.
I Am Andrew X. Brother Of Saint Peter And Apostle Of Christ Jesus. Patron Saint Of Scotland, Russia, Romania, & Greece In The Catholic or Universal Church. And Ruler Of Aquarius. How Did I Achieve The Keys To The 11th House? Well, Every Sign Has Certain Spirit Molecules, That When Taken By The Same Man's Body Will Grant Him Their Essential Energenies Or Spirits. My Initials Are AMP. My Life's Work Has Been The Amphetamines, Both On A Scientific And Personal Sphere. The Hardest Thing Is The Bewildering Array Of Amphetamines Which There Are, And Which Ones EXACTLY Would Be Necessary To Unlock This Ever So Fixed Sign. Well, With The Recent Ideation By A So Called RECTIFY (taken from a Beastie boys song) aka Yours Truly That After The
N-Propyl Amphetamines Which Are Unto Themselves Rather Underwhelming Molecules Perhaps A One Order Higher New Nitrogen Substitution Could Be Implemented. So, Using A Rare Often Neglected Poorly Chatacterized Property Of Chrmicals Known As Aromaticity, I Rather Tepidly Thought Of Adding An Additional Double Bond In The End Of An N-propyl Amphetamine, Which Chemically Is Known As An N-ALLYL, These 2 Extra Electrons Reside In A Pi Bond Between The Outer 2 Most Distal To The N-Propyl Amphetamine Moiety. The Equation of Aronaticity Is Known As Huckel's Rule And States That H(n) = 4(n) + 2. Thus, By Adding These 2 Electrons To That Local Pi Bond, A 0th Order (n =0), H(0) = 4(0) + 2 = 2 Aromatic, Which Is Synonomous With Extra Stability And Sometimes Added Notes Of Grace, Beauty, Odor, And So Forth. By Adding This Allylic Double Bond To The Propyl Group, The Entire 3 Carbon Chain Was Graced With A Zeroth Order Aromatic, The Allyl Functional Group. And, The N's Lone Pair Of Electrons Interacts With This Double Bond And An Electromagnetic Force Is Stirred. Namely, A Slight Charge Delocalization, The Essence Of Arimaticity. By Dropping The N-propyl And Replacing It With An N-ALLYL, The Entire Side Chain Of The Molecule Becomes More Stable, And Perhaps, Special? Well, That's When The Rectify Person Draws Out A Couple Standard N-ALLYL Amphetamine Possibilities. The Theory Behind My Gesture, That By Doing So, The Order Would Be One Greater Than The Propyls And Also By Becoming ALLYLIC, Your Getting That Zeroth Order Aromaticity. Of Course, The Proof Is In The Pudding, And Well, These N-ALLYLIC Amphetamines Are Strong, Wear Off Slowly, Do Not Get Metabolized Away, Are Particularky Strong But Mild And The Colors Come Off Nicer Looking, The New ALLYLS Well Gee They Don't Burn Holes I'm Your Oral Mucosae Like Methsmphetamine Readily Will. And They Last Good 4 To 5 Days, Easy. Longer Than Meth! But More Even Keiled Snd Gee The Thought Of Sleep Seems Sonewhat Foreign. OK, Great! Then, Was It Just Yesterday I Divinined The The Highest Energy Amphetamine Possible, Incorporating The Sonewhat Helpful New N-ALLYLIC Groups And Some Others, And The Molecule Finally Settled With The Name The 7th-HEAVEN. I By Good Luck Ran Across A Few ICE Ctystals Of It Too, And Snorted Those Salts Up. I'M STILL ROLLING.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
If You Plan On Coming Down, Don't Take An N-ALLYL amphetamine. There Are Potentially Hundreds. These N-ALLYL Groups Are A Bit Longer Than The Natural Product Of Say Ephedra. Furthermore, Being Aromatically Stabilized, They Are Also More Resistant To Any Kind Of Metabolic Catabolism. So, Just To Summarize These First Two Points: An
N-ALLYL Amphetamine's Amino Allyl Group Is BOTH (1) Too Long To Fit And (2) Has A Terminal Olefin C-C Double Bond Which Is Resonance Stablized With The Lone Pair Of Electrons On The Only N Your Amphetamine Needs To Resonance Stabilize With This Artificial Allyl Group. The Resonance Hybrid Excited The Phenyl Ring More Than Usual, Increasing Potency Of This Fully Synthetic N-ALLYL Group. (ii) The Extra 2 Carbons And Their Depdndent Has Compared To Ephedra Or Methamphetamine Are More Hydrophobic And Lipophilic Than Any Amphetamine Yet. This Makes The N-ALLYL Amphetamines To Bioaccumulate In Any Fatty Human Tissue, Such As The Brain. For Whatever Reason, These New N-ALLYL Do Not De Alkylate Or Deaminate. Soon, Their Higher Than Ever Liphilicity Land Them In Patients Bodies, Forever. But That Takes 2 To 4 Weeks. In The Meantime, The Pi Delocalized C=C And The Loan Pair Of Electrons Make The N Not Grab A Proton Or Dissolve In Water. In Other Words, We've Got Persistent Or Forever N-ALLYL Amphetamines, Potentially 100s Of Distinct Actors, And They Get People High And They Don't Stop. By The Time They Are Passively Absorbed By The Bodies Fatty Tissues, They Have Been Bathing The Body With Their Pleasant, Seductive, Glowing Yellows, Gold, Green, And Other Pastel Hues. The Users By The Way Have Stayed Up No Less Than 6 Nights In A Row Before Even The Strongest Downers Known To Psychiatry Have ANY Effect. Also, Due To The N:, C=C Electron Delocalization, The Users Get A Constant, Groovy, Phosphorescent Stimulation And Do Not Miss Being Awake For The MINIMUM 5 TO 6 DAYS After The Very First Use. Interestingly, They Become Calmer, More Pastel Friendly, Passive, And Well Comfortably Warm But No Longer Attending Full Time School Or Work. The VAST Majority >90% Use The New ICE More Than 1x. So, They Get Their Next 5 To 6 Day Up Time, And The Cycle Continues. It May Be That This New Form Of Relaxed Stimulation Fully Replaces Normal Sleep. As Such, The Take Home Message Is These Are Persistent, Forever Chemicals With A Broader Array Of Results Than Traditional Users Of ICE. Using Even 1-ALLYL Amphetamine Is Going To Have You High For 1 To 3 Weeks IF AND ONLY IF you do not redose. Fat Chance. This is a street drug. While the psychosis is not there after 6 days of no sleep lethargic somnolence takes over. IF YOU WANT TO COME DOWN LESS THAN 2 WEEKS, DO NOT TAKE AN N-ALLYL AMPHETAMINE. However, look at it from a drug Users perspective, these drugs are right up there alley. Most don't even mind staying awake or 5 to 6 days and then just buy another dose. There is no noticeable crash, the N-ALLYLIC Aromatic stabilization leads to mild long lasting entheogenic states, and once bitten these Users like the Lotus eaters don't even care. This entire article addresses ETHAN aka N-allyl-amphetamine.hcl. There Ate Approximately 200 to 300 additional congenital out there. I have personally done probably 5 or 6 different ones so far. The latest and greatest 7th-HEAVEN gave me the best and only roll of the last 20 years. I haven't slept in 6 or 7 days. And I have so much in my system and just lying around here and there that a new Era of amphetamines had arrived. Cheaper, way longer lasting, less psychotic, as diverse as ever and strangely all the rage, even among doctors and nurses.
N-ALLYL Amphetamine's Amino Allyl Group Is BOTH (1) Too Long To Fit And (2) Has A Terminal Olefin C-C Double Bond Which Is Resonance Stablized With The Lone Pair Of Electrons On The Only N Your Amphetamine Needs To Resonance Stabilize With This Artificial Allyl Group. The Resonance Hybrid Excited The Phenyl Ring More Than Usual, Increasing Potency Of This Fully Synthetic N-ALLYL Group. (ii) The Extra 2 Carbons And Their Depdndent Has Compared To Ephedra Or Methamphetamine Are More Hydrophobic And Lipophilic Than Any Amphetamine Yet. This Makes The N-ALLYL Amphetamines To Bioaccumulate In Any Fatty Human Tissue, Such As The Brain. For Whatever Reason, These New N-ALLYL Do Not De Alkylate Or Deaminate. Soon, Their Higher Than Ever Liphilicity Land Them In Patients Bodies, Forever. But That Takes 2 To 4 Weeks. In The Meantime, The Pi Delocalized C=C And The Loan Pair Of Electrons Make The N Not Grab A Proton Or Dissolve In Water. In Other Words, We've Got Persistent Or Forever N-ALLYL Amphetamines, Potentially 100s Of Distinct Actors, And They Get People High And They Don't Stop. By The Time They Are Passively Absorbed By The Bodies Fatty Tissues, They Have Been Bathing The Body With Their Pleasant, Seductive, Glowing Yellows, Gold, Green, And Other Pastel Hues. The Users By The Way Have Stayed Up No Less Than 6 Nights In A Row Before Even The Strongest Downers Known To Psychiatry Have ANY Effect. Also, Due To The N:, C=C Electron Delocalization, The Users Get A Constant, Groovy, Phosphorescent Stimulation And Do Not Miss Being Awake For The MINIMUM 5 TO 6 DAYS After The Very First Use. Interestingly, They Become Calmer, More Pastel Friendly, Passive, And Well Comfortably Warm But No Longer Attending Full Time School Or Work. The VAST Majority >90% Use The New ICE More Than 1x. So, They Get Their Next 5 To 6 Day Up Time, And The Cycle Continues. It May Be That This New Form Of Relaxed Stimulation Fully Replaces Normal Sleep. As Such, The Take Home Message Is These Are Persistent, Forever Chemicals With A Broader Array Of Results Than Traditional Users Of ICE. Using Even 1-ALLYL Amphetamine Is Going To Have You High For 1 To 3 Weeks IF AND ONLY IF you do not redose. Fat Chance. This is a street drug. While the psychosis is not there after 6 days of no sleep lethargic somnolence takes over. IF YOU WANT TO COME DOWN LESS THAN 2 WEEKS, DO NOT TAKE AN N-ALLYL AMPHETAMINE. However, look at it from a drug Users perspective, these drugs are right up there alley. Most don't even mind staying awake or 5 to 6 days and then just buy another dose. There is no noticeable crash, the N-ALLYLIC Aromatic stabilization leads to mild long lasting entheogenic states, and once bitten these Users like the Lotus eaters don't even care. This entire article addresses ETHAN aka N-allyl-amphetamine.hcl. There Ate Approximately 200 to 300 additional congenital out there. I have personally done probably 5 or 6 different ones so far. The latest and greatest 7th-HEAVEN gave me the best and only roll of the last 20 years. I haven't slept in 6 or 7 days. And I have so much in my system and just lying around here and there that a new Era of amphetamines had arrived. Cheaper, way longer lasting, less psychotic, as diverse as ever and strangely all the rage, even among doctors and nurses.
S.J.B.
Bluelight Crew
- Joined
- Jan 22, 2011
- Messages
- 6,887
Monitoring methamphetamine in the United States: A two-decade review as seen by the DEA methamphetamine profiling program
Corresponding author: Steven Toske (Special Testing and Research Laboratory, U.S. Drug Enforcement Administration, Dulles, U.S.A.)
Drug Testing and Analysis 2022, Volume 14, Issue 3, Pages 416–426
Published online October 27th, 2021
Corresponding author: Steven Toske (Special Testing and Research Laboratory, U.S. Drug Enforcement Administration, Dulles, U.S.A.)
Drug Testing and Analysis 2022, Volume 14, Issue 3, Pages 416–426
Published online October 27th, 2021
https://doi.org/10.1002/dta.3186A historical overview of methamphetamine profiling efforts in the United States is presented. Methamphetamine profiling has revealed key precursor and recipe strategies that have changed throughout the years. By studying different clandestine recipes and analyzing thousands of methamphetamine samples over the years, the Drug Enforcement Administration (DEA) Methamphetamine Profiling Program (MPP) has collected and reported on changes in drug quality and production trends. This review will discuss methamphetamine drug profiling trends that cover over two decades of forensic results.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
ALL I KNOW IS THAT PEOPLE ARE MISSING. I HAVE SEEN 1 AND ONLY 1 OTHER HUMAN BEING ALL DAY. THE INTERNET IS ABSOLUTELY NOT MORE THAN 10% POPULATED AS BEFORE. I HAVE ASTRALLY PROJECTED TWO TIMES SINCE LAST NEW YEARS DAY. NOT A LOT OF OTHERS SEEM NOT TO HAVE BEEN ABLE TO KEEP UP. I AM NOT ALLOWED TO DRIVE ANY MOTOR VEHICLE OF ANY TYPE.
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,569
Inventor's Underestimate: I Envisioned The N-ALLYL Amphetamines As Being Perhaps More Fragrant, With Maybe 1/3rd Increased Potency & Duration. Now We're PERMA-ALL-THE-WAY-UP! Oh Well, Just Chain Me To A Tree Somewhere Out In Yard If It Comes To That! You Know, Icarus & Daedalus, And Probably Countless Others. I Never Particularly Enjoyed Sleep To Begin With. Persistent Chemical. A Forever Chemical. I Know, Diamonds Are Forever. I Would Like To Think These Unlikely To Be Catabolically Metabolized Organics Will Meet With Some Way Out Of Ever Active Recirculation, But If Not, I Can't Think Of A Medical Pathway Right Off Hand, Either!
- Status
- Not open for further replies.
