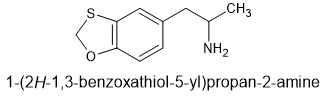
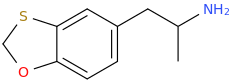
Good question.
1,3-Benzoxathiole is commercially available. From their, tri(o-tolyl)phosphine + tributyl tin methoxide + isopropyl acetate + PdCl2 will result in a mixture of the BTPCP and an unwanted (ortho) positional isomer.
Exactly the same way 6-APB was made.
I presume the above is a name reaction, but I didn't search, I just checked that their WAS a facile route.
You will note that I placed the S at the meta position rather than the para. I HAD thought that it was the S at the para that was the issue but fast&bulbous, I'm sure, knows more than me.
The funny thing is that the 5/6 APB synthesis is in a patent! No RC vendor 'invented' it, they just undertook a lot of datamining. Nothing wrong with that EXCEPT when it's presented as new work.