I can assure you that pyeyzolam really does accurately reproduce the positive effects of alcohol
-Anxiolytic
-Increased sociability
-Improved mood
-Non-vascular muscle relaxant
Without the negative effects of alcohol
-Disinhibition
-Loss of judgement
-Anterograde amnesia
-Mood lability
-Ataxia
-Nausea/Emesis
-Physical dependence
As well as the physical damage of alcohol which in it's acute phase we recognize as a hangover but in it's chronic phase includes Korsakov syndrome, fatty liver disease, heart disease, kidney disease and so on.
It turns out that alcohol primarily acts on the a1 and a5 subunits of the GABA receptor and it's the a1 activity that causes most of the negative symptoms of alcohol (think Z-drugs which are all a1 selective) and a5 activity that produces all of the positives (unless people WANT disinhibition, loss of judgement and/or amnesia, I suppose). We know how to simulate the good parts of alcohol and understand why their are positive and negative effects.
No, I'm 100% sure they have at least 3 because I sold pyeyzolam to Alcarelle (I was offered cash or shares and took cash because had no faith in their business model - too many questions they had no answers for). BUT if pyeyzolam is mixed with alcohol, it multiplies (not adds) to the effects of the alcohol so while 25mg of pyeyzolam is more or less like drinking a bottle of wine. If you drink a bottle of wine after 25mg of pyeyzolam you WILL black out and that is dangerous.
Their is no getting around that.
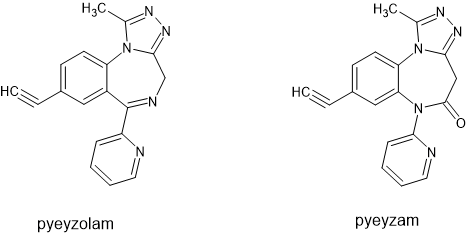
The only failing of pyeyzolam was it's dose-response curve. Up to about 20mg it does nothing and then from 20-30mg it goes from a glass of wine to two bottles of wine. BUT we discovered why and tested it. Alcohol binds to the a1 & a5 subunits at the junction of the b1, b2 and b3 subunits and so you need a candidate that isn't beta selective. Step one was to have pyeyzam (the 1,5 analogue of pyeyzolam) made and tested and alone it wasn't very active. It took 40mg for anything to happen and then the same glass of wine level BUT it had a plateau in it's effects. However much you take (and we did take a lot) it only ever got to the same as around two or three glasses of wine.
BUT if you mix pyeyzolam and pyeyzam in the appropriate proportions, you end up with a product that will emulate anything from a glass of wine to a bottle of vodka.
Image zam hosted in ImgBB

ibb.co
So the problem of emulating alcohol is solved. As people may know, like pyrazolam both pyeyzolam and pyeyzam are excreted by the body unchanged. This was a FEATURE of the design. Medicinal chemistry is littered with medicines that a small proportion of the population metabolized in a different manner and produced toxic metabolites. The most famous is the antibiotic Trovan (trovafloxacin) which Pfizer trialled on children in Africa and turned into a disaster. But their are actually many more - it's just that pharmaceutical giants are able to cover up most cases. So a medicine that isn't metabolized has a distinct advantage.
But the problem with the alcohol mimic I mention is that it requires TWO drugs. Pyeyzolam is a5b1y2 selective while pyeyzam binds to both a5b2y2 and a5b3y2. So BOTH of them would have to go through testing essentially doubling the cost (I've worked with Huntingdon Life Sciences and so I do know how medicines are tested).
The breakthrough I made (and won't be sharing publicly) is that it's possible to produce something in-between pyeyzolam and pyeyzam that isn't beta selective. Now the modelling of ALL the different subtypes of the benzodiazepine sites was carried out by one James T. Cook at UWM in the US.
Above is a VERY good resource. Cook essentially spent 30+ years researching the benzodiazepine site and almost all of his students worked on 3DQSAR analysis.
I wouldn't say my single-drug solution is some great feat of medicinal chemistry but rather a bit of lateral thinking. Tautomers and more specifically mesoionic compounds hadn't been explored but it's possible to design a tautomer that places the pendent aromatic in the same position as a 1,4 benzodiazepine and in the same position as a 1,5-benzodiazepine.
I was kind of tipped off by looking into French research into 4-phenyl-4H-benzo[d][1,3]oxazin-2-amines (such as Etifoxine). I couldn't work out why the N-ethyl was the most active by a HUGE margin until I realized that of course, it's a tautomer.
Frankly, I believe the most facile path is via China. Their one party system certainly has it's failings but if an existing production/distribution/retail/marketing/legal framework can be adapted for a new product (I mean the alcoholic drinks industry) then it seems to me they can do it. Jobs, taxation and duty are, in truth, more important to politicians than safety so if overnight the ethanol were replaced by a safe mimic, essentially nothing would have to change apart from the list of ingredients on the packaging.
Sorry to bang on about it but GABA Labs seem to think they can begin begin as a niche product. I totally disagree. I think it needs a government to DECIDE that alcohol is dangerous and impose
a change. Not possible in a democratic nation. InBev and the other massive brewing companies will fight to keep alcohol as they are defending a market share.