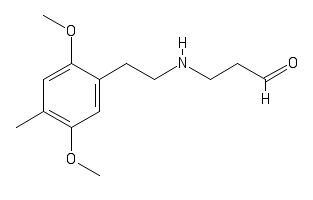
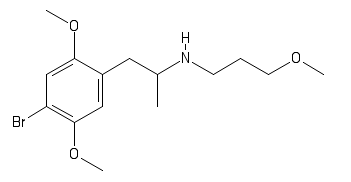
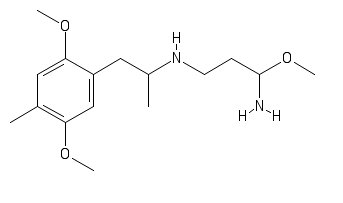
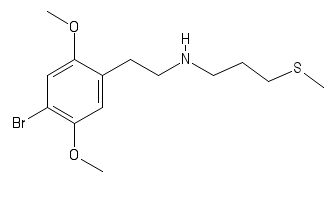
polymath
Bluelight Crew
It's quite surprising that these aliphatic sympathomimetics can produce a cocaine like discriminative stimulus in experiments
 www.ncbi.nlm.nih.gov
www.ncbi.nlm.nih.gov
But ephedrine does, too, so it doesn't mean it's any good as a drug.
Abuse Liability of the Dietary Supplement Dimethylamylamine - PMC
Dimethylamylamine (DMAA) is a component of many dietary supplements and has recently been associated with numerous adverse effects, prompting the US military and World Anti-Doping Agency to ban its use as a supplement. The current study aimed to ...
In the present study, DMAA produced locomotor depression, fully substituted for the discriminative stimulus effects of cocaine, produced partial substitution for methamphetamine (77% drug-appropriate responding), and produced a conditioned place preference. The potency of DMAA was comparable in all three assays (between 2.3–3.3 mg/kg). These results indicate that DMAA produces reward-like effects and may produce subjective effects similar to that of abused psychostimulants, and therefore has potential for abuse.
But ephedrine does, too, so it doesn't mean it's any good as a drug.