Neuropharmacology of DMT
Theresa M. Carbonaro, Michael B. Gatch
N,N-Dimethyltryptamine (DMT) is an indole alkaloid widely found in plants and animals. It is best known for producing brief and intense psychedelic effects when ingested. Increasing evidence suggests that endogenous DMT plays important roles for a number of processes in the periphery and central nervous system, and may act as a neurotransmitter. This paper reviews the current literature of both the recreational use of DMT and its potential roles as an endogenous neurotransmitter. Pharmacokinetics, mechanisms of action in the periphery and central nervous system, clinical uses and adverse effects are also reviewed. DMT appears to have limited neurotoxicity and other adverse effects except for intense cardiovascular effects when administered intravenously in large doses. Because of its role in nervous system signaling, DMT may be a useful experimental tool in exploring how brain works, and may also be a useful clinical tool for treatment of anxiety and psychosis.
Introduction
N,N-dimethyltryptamine (DMT) is an indole alkaloid widely found in nature. It is an endogenous compound in animals and in a wide variety of plants found around the globe. Major plant genera containing DMT include Phalaris, Delosperma, Acacia, Desmodium, Mimosa, Virola, and Psychotria, but DMT has been found even in apparently innocuous sources, such as leaves of citrus plants, and in the leaves, seeds, and inner bark of mimosa tenuiflora, which has become a source of livestock poisoning.
DMT has become of interest because when ingested, it causes brief, episodic visual hallucinations at high concentrations. DMT is one of the major psychoactive compounds found in various shamanistic compounds (e.g., ayahuasca, hoasca, yage) used in South America for centuries and has, more recently found its way into Europe and North America as a recreational drug.
Recreational use of DMT
Most hallucinogens such as lysergic acid diethylamide (LSD) and 2,5-dimethoxy-4-methylamphetamine (DOM) cause sensory distortion, depersonalization at high doses, and at least one (N,N-Diisopropyltryptamine, DiPT) causes auditory distortions, whereas some compounds such as DMT (found in ayahuasca), psilocybin (mushrooms) or mescaline (peyote) cause episodic visual effects. In the late 1990s, Rick Strassman conducted the first human research with hallucinogens in 20 years, examining the physiological effects and self-reports from people receiving DMT in carefully controlled settings. A book describing these results was published in the popular press. Strassman concluded that DMT is a powerful tool for self-discovery and understanding consciousness, which may have helped to drive interest in recreational use of DMT and related tryptamine hallucinogens. In recent years, recreational use of DMT has been increasing; for example, Cakic et al., reported that 31% of recreational DMT users endorse psychotherapeutic benefits as the main reason for consumption. Similar to ayahuasca, recreational users have made similar concoctions referred to as pharmahuasca. These are of capsules containing free-base DMT and some monoamine oxidase inhibitors (MAOI) such as synthetic harmaline or Syrian Rue.
It is unclear what proportion of users of hallucinogenic tryptamines have adverse events serious enough for hospitalization, but it seems that the synthetic hallucinogenic compounds, such as 25I-NBOMe may be more dangerous than the plant-derived compounds. Databases derived from Poison Control and Emergency Department visits only sparing differentiate between hallucinogenic compounds taken and lack adequate records of DMT-specific cases. Street drugs mostly contain powdered DMT, whereas ayahuasca also contains harmine-related compounds, which limit toxic effects. However, aside from the acute cardiovascular effects there have been no consistent reports of toxic effects of long-term use of DMT in the literature. In fact, there has been a report that DMT is neuroprotective. Without more data on the recreational use of this class of compounds, it is not possible to conclude whether the synthetic hallucinogens are indeed more toxic or whether the social context may contribute to the effects.
It is likely that most adverse effects of hallucinogens are psychological effects, such as intense fear, paranoia, anxiety, grief, and depression, that can result in putting the user or others in physical harm or danger. Anecdotal reports describe psychologically challenging experiences with DMT and other psychedelic compounds. The rates of occurrence for these effects have not been properly accounted for. However, in the case of psilocybin, about 30% of laboratory experiences include psychologically challenging experiences. Even though DMT may not produce physical toxicity, severe psychological adverse effects can occur.
Endogenous roles of DMT
Although widespread biological presence of DMT is acknowledged, the biological function of DMT remains a mystery. DMT is found in low concentrations in brain tissue. DMT concentrations can be localized and elevated in certain instances, for example, DMT production increases in rodent brain under stress. Formerly, endogenous DMT was thought to exist at concentrations too low to produce pharmacological effects, but two discoveries changed that. First, trace amine-associated receptors (TAAR) are activated by DMT and other molecules, and second, DMT can be locally sequestered in neurotransmitter storage vesicles at pharmacologically relevant concentrations, thereby being able to active other pharmacological receptors, e.g. serotonin. These findings suggest that DMT may have a role in normal physiological and/or psychopathology. What that role may be has not yet been established.
Although the serotonin system has been thought to be the main contributor to the psychedelic effects of DMT, other behavioral effects have been observed which do not involve the serotonin or other monoaminergic systems; such as jerking, retropulsion, and tremors. In addition, molecular effects of DMT have been identified that are not mediated by serotonin receptors. For example, DMT-enhanced phosphatidylinositol production is not blocked by 5-HT2A receptor antagonists. More recent hypotheses for molecular roles of endogenous DMT have developed over the last decade, and include the potential involvement of TAAR and sigma-1 receptors. Interactions of both TAAR and sigma-1 receptors will be discussed in detail in subsequent sections.
There has been a great deal of speculation about the role of DMT in naturally occurring altered states of consciousness, such as psychosis, dreams, creativity, imagination, religious and/or spiritual phenomena, and near-death experiences. Additionally, DMT may play a role in waking reality. Waking reality is created in a similar way to altered states except that the normal state correlates with event in the “physical” world. Thus, waking reality can be thought of as a tightly regulated psychedelic experience and altered states arise when this regulation is loosened in some fashion. This model predicts that the sensory-altering effects of administered psychedelics are a result of the compound acting directly via neuropharmacological mechanisms in regions of the CNS involved in sensory perception. More simply, DMT may potentially act as a neurotransmitter to exert a signaling function in regions of the CNS, which are involved in sensory perception.
Other theories propose that DMT may be important in psychiatric disorders. Data from early studies of DMT suggested that DMT may be a schizotoxin, and various authors hypothesized that DMT was a key factor in causing schizophrenia. This hypothesis is no longer accepted, but it is still thought that DMT may play a role in psychotic symptoms. Similarly, DMT was thought to be neurotoxic, but more recent research suggests that DMT may actually be neuroprotective.
More recently, Jacob and Presti proposed that endogenous DMT may have an anxiolytic role based on the reported subjective effects of DMT administered in low doses, which would result comparable concentrations and biological actions to those of endogenous DMT. Sensory alterations commonly described by people taking DMT occur only when relatively high concentrations of DMT are administered. These high concentrations are similar to those observed in the synapse when endogenous DMT is released.
The putative roles of DMT will be explored in more detail in subsequent sections of this review. The review will begin by addressing the basic mechanisms of action of DMT, both pharmacokinetic and pharmacodynamic. It will then examine evidence regarding the neuropharmacological effects of DMT, from both behavioral studies of the exogenous effects of DMT, and from molecular studies of sites of action of endogenous DMT. Next, the review will turn to the use of DMT both as a model for various disorders and the use of DMT to treat some of these disorders. The review will conclude with the effects of DMT on other organ systems besides the central nervous system.
Pharmacokinetics of DMT
Intravenous administration of radio-labeled DMT in rabbits produces entry into the brain within 10 s and excretion via the kidneys, such that no traces of DMT or metabolite was measured in urine 24 h post administration. However, DMT could still be detected at 2 and 7 days (0.1% of initial dose) post administration. In the same study, tryptamine was eliminated within 10 min. These findings show that even after complete clearance of a dose of DMT from the blood, DMT is still present in the CNS, and imply that DMT is being produced in the CNS. The subjective effects of intravenous administration of DMT peak at about 5 min and are gone by 30 min. Intramuscular effects of DMT hydrochloride or DMT fumarate have a rapid onset within 2 – 5 min and can last 30 - 60 min, and the effects are generally less intense than intravenous or inhalation routes of administration. The hallucinogenic effects of DMT in the formulation of ayahuasca generally appear within 60 min, peak at 90 min and can last for approximately 4 h. Typical doses of smoked or inhaled free-base DMT are 40 - 50 mg, although dose may be as high as 100 mg. The onset of these doses of smoked DMT is rapid, similar to that of i.v. administration, but lasts less than 30 min. Smoked DMT effects are extremely intense. Intranasal free-base DMT was inactive, as was DMT administered rectally.
To establish that DMT acts as a neurotransmitter rather than merely being a by-product of the metabolism of other bioactive molecules, it is necessary to establish that it is synthesized, stored, and released. It is of interest that DMT can pass through three barriers with the help of three different mechanisms so that it can be compartmentalized and stored with the brain. These three mechanisms may yield high intracellular and vesicular concentrations within neurons, which suggests that DMT may have a biological role. Processes for the transport of glucose and amino acids are given similar biological priority, which may suggest that DMT is present in the body for more than its psychedelic effects, such as an adaptive role in biological processes, or a universal role in cellular protective mechanisms.
Synthesis
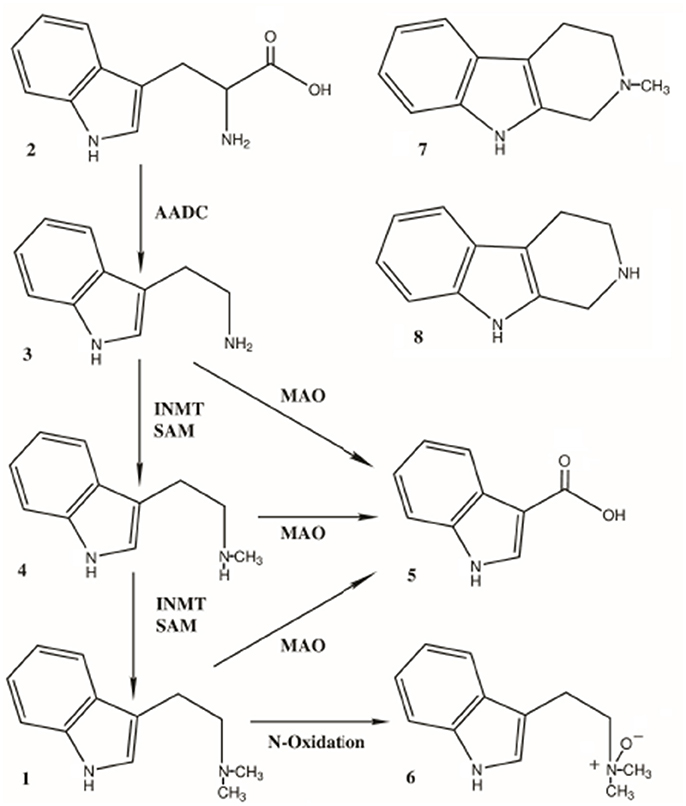
Endogenous DMT is synthesized from the essential amino acid tryptophan, which is decarboxylated to tryptamine. Tryptamine is then transmethylated by the enzyme indolethylamine-N-methyltransferase (INMT), which catalyzes the addition of methyl groups resulting in the production of NMT and DMT. NMT can also act as a substrate for INMT-dependent DMT biosynthesis. INMT is widely expressed in the body, primarily in peripheral tissue such as the lungs, thyroid and adrenal gland. INMT is located in intermediate levels in placenta, skeletal muscle, heart, small intestine, stomach, retina, pancreas, and lymph nodes. It is densely located in the anterior horn of the spinal cord. Within the human brain, highest INMT activity has been found in uncus, medulla, amygdala, frontal cortex, and in the fronto-parietal and temporal lobes. Cozzi et al. has shown INMT is also located in the pineal gland. Based on rodent brain cellular fractionation studies 70% of INMT activity is found in the supernatant and 20% in the synaptosomal fractions, suggesting the enzyme is located in the soma of cells, which are fractured during the homogenization process. The wide distribution of INMT implies a wide distribution for DMT.
The enzymatic activity of INMT is closely regulated by endogenous inhibitors. DMT at high concentrations (10-4 M) yields a 90% inhibition of rabbit lung INMT. In addition, the same tissues that contain INMT also contain enzymes that metabolize DMT. Only a small fraction of DMT made intracellularly is actually released into the blood. This process helps explain the inconsistent detection levels assessed in many studies discussed below. DMT production is increased under stress in rodent brain and adrenal gland. Whether the stress-induced mechanism for increasing DMT is due to increasing INMT activity, or a decrease in DMT metabolism remains unknown.
Accumulation and storage
As previously mentioned, it has been hypothesized that high, local concentrations of DMT can occur within neurons and potentially widely produced in peripheral organs, especially in the lungs. Frecska and colleagues summarized a three-step process by which DMT is accumulated and stored. In step 1, DMT crosses the blood brain barrier by active transport across the endothelial plasma membrane, which is accomplished via Mg2+ and ATP-dependent uptake. In step 2, uptake of DMT into neuronal cells is accomplished via serotonin uptake transporters (SERT) on neuronal plasma membrane. In step 3, facilitated sequestration of DMT into synaptic vesicles from the cytoplasm is accomplished by the neuronal vesicle monoamine transporter 2). DMT inhibited radiolabeled 5-HT uptake via the serotonin transporter (SERT) and VMAT2 with Ki values of 4 and 93 μM, respectively. DMT that has been taken up and stored within cells via SERT and VMAT2 and exhibit high binding-to-uptake ratios, >11 for SERT and >10 for VMAT2. High binding ratios suggest that there are separate substrate and inhibitor sites for SERT and VMAT2 and further supports that DMT (and other tryptamines) are substrates for both transporters.
The high levels of DMT concentration found in vesicles are needed for various pharmacological actions including activation of sigma-1 receptors and TAARs as described below. Once uptake and storage of DMT has been completed, it can remained stored in vesicles for at least 1 week and can be released under appropriate stimuli. Through these three steps, peripheral synthesis of DMT, consumption of DMT-containing plant matter, or systemic administration of DMT can influence central nervous system functions.
Bioavailability of exogenous DMT
DMT is not orally active. This is likely due to rapid degradation by peripheral monoamine oxidase (MAO), the enzyme responsible for catalyzing the oxidative deamination of endogenous biogenic amines. For hallucinogenic or psychedelic phenomena to occur, plasma concentration must be between 12 – 90 μg/L with an apparent volume of distribution of 36-55 L/kg, which roughly corresponds to a plasma concentration of 0.06 – 0.50 μM. In order for DMT to be bioavailable, oral formulations such as ayahuasca contain Banisteriopsis caapi and other beta-carboline harmala alkaloids that act as MAOIs. MAO-A inhibitors such as iproniazid prolongs the half-life of DMT in rat brain, and can extend the time course effects of DMT in drug discrimination from 30 min to 60 min. Exogenous DMT formulations containing a reversible MAOI (such as ayahuasca) can result in blood levels up to 1.0 mg/ml or higher. On average a 100 mL dose of ayahuasca contains about 24 mg of DMT. Interestingly, DMT is itself a short-acting monoamine oxidase inhibitor at high doses, and is selective for MAO-A. In these studies, DMT decreased serotonin and dopamine deamination in rat striatum concomitantly with rapid onset. Normalization occurred 2 hours later with an ED50 of 25 mg/kg for degradation of both serotonin and dopamine.
Degradation and elimination
A small fraction of exogenous DMT is excreted in urine as the parent compound. DMT is primarily metabolized by MAO, although there is evidence that DMT can also be metabolized by peroxidases, leading to a variety of other metabolites. In both human and rodent models, none of the metabolites produced DMT-like effects. From the MAO pathway, several indole compounds are produced: NMT, 6-OH-DMT, 6-OH-DMT-NO, DMT-NO, and IAA. The major metabolites of DMT are DMT-NO, and IAA. In rabbit liver microsomal fractions pretreated with MAOI iproniazid, five indole compounds were found: DMT, NMT, 6-OH-DMT, 6-OH-DMT-NO, and DMT-NO; however, in rabbit brain microsomal preparation again with iproniazid pretreatment, no 6-hydroxy metabolites were identified. These findings suggest that metabolism of DMT is somewhat different in brain and in the periphery.
Formation of IAA was thought to be likely due to oxidase deamination of NMT, but was later established to be also in part by direct deamination by MAO. Pretreatment with the MAO-I iproniazid in rat whole brain homogenates inhibited IAA formation by 83%, NMT and DMT-NO formation were inhibited by 90%, suggesting that the increase in behavioral half-life of DMT is due to MAO-inhibition and inhibition of the enzymes responsible for demethylation and N-oxidation as well.
Erspamer first recorded IAA as a metabolite of systemically administered DMT in rodent urine, which represented less than 3% of the injected dose of DMT. In human volunteers, 8.3% of the administered dose of DMT was recovered as IAA. Around 50% was recovered as IAA but also as DMT-NO and other MAO-independent compounds. Neither the Erspamer nor Szara study detected unchanged DMT in urine. Little DMT is found unchanged in the urine of ayahuasca users despite taking it with harmala alkaloids. In another study in humans, 0.16% of DMT was recovered as DMT following a 24-hour urine collection. In both of these studies, DMT concentration peaked in blood within 10-15 minutes and was essentially undetectable by one hour. Approximately only 1.8% of the injected dose was present in blood at any one time.
Oxidative deamination of DMT by MAO may not be the sole metabolic pathway in humans. A study by Gomes et al. suggests that a different metabolic pathway by which DMT can be oxidized by peroxidases may be responsible for increasing cytotoxic activity of peripheral-blood mononuclear cells. Metabolites in this pathway include hydroxy-DMT, N,N-dimethyl-N-formyl-kynuramine, and N,N-dimethyl-kynuramine. Barker et al. suggest other possible metabolites of DMT include 1,2,3,4-tetrahydro-beta-carboline (THBC) and 2-methyl-THBC.
Detection of endogenous DMT in blood, urine, and cerebrospinal fluid
DMT as an endogenous compound can be measured in human body fluids, including blood, urine and cerebral spinal fluid. Levels of endogenous DMT do not appear to be regulated by diet or gut bacteria. Infrequent and inadequate sampling methods used over time make it difficult to determine specific details pertaining to DMT production in the body. For example, we still do not know if DMT is produced in phasic or diurnal cycles. Measureable concentrations seem to only occur intermittently, and exact tissue source or sources of DMT is still unclear. It is commonly thought that the adrenal gland and lungs are the most common places for the highest amount of DMT production, since this is where highest levels of INMT have been reported.
A review by Barker assessed 69 studies that reported endogenous DMT detection and quantities reported in urine, blood, and cerebrospinal fluid, primarily comparing detection levels within healthy controls and schizophrenic patients. DMT in urine was examined in 861 individuals (635 patients), 276 patients and 145 controls were positive for DMT. Throughout the studies, there were inconsistent sampling methods, including various of amounts of urine used in assays, and a range of techniques and analytical approaches were used. Some studies took dietary influences into consideration, but found no associations with endogenous DMT levels. Inconsistent units of measurement were also used across studies. Concentrations in urine range from 0.02 to 42.98 +/-8.6 (SD) ug/24h, and from 0.16 to 19 ng/ml. In blood, data from 417 (300 patients) individuals were examined, 44 patients and 28 controls were positive for DMT. One study was responsible for 137 of the negative samples. Like detection in urine, extraction methods and analytical approaches were highly inconsistent. Testing procedures included discrepancies of samples coming from plasma, serum and/or whole blood, while others had limit detections of 0.2 DMT/ml. Higher concentrations of DMT are extracted from whole blood compared to plasma, but there is no difference in venous and arterial blood. When concentrations were reported, not just whether it was present or not present, it ranged from 51 pg/ml to 55 ng/ml. DMT was detected in cerebrospinal fluid in 4 studies, which tested 136 individuals (82 patients). Of those, 34 patients and 22 controls were positive for DMT. Concentrations ranged from 0.12 to 100 ng/ml. DMT can be detected as an endogenous compound in urine, blood, and cerebrospinal fluid. Even with inconsistent detection methods, DMT does not appear to be related to the onset of schizophrenia, since it seems to be detected more so in healthy controls compared to patients.
Clinical effects
Oral dosing of DMT via ayahuasca produces both behavioral and neurochemical effects, such as decreases in motor activity, impairment of cognitive function, sympathomimetic effects, increased prolactin and cortisol levels, and decreased lymphocytes increased natural killer cells. Doses of ayahuasca 15 or 30-fold higher than commonly used ritual doses increased serotonergic neurotransmission. Long-term use of DMT in ayahuasca produces measurable brain changes. Long-term ayahuasca users show difference in midline brain structures using MRI versus matched controls. Interestingly, whereas ayahuasca produced modest impairment of cognitive function in inexperienced users, little or no impairment was observed in experienced users.
Tolerance
Several early studies demonstrated that DMT does not produce tolerance. When DMT was administered to squirrel monkeys for 36-38 days, it failed to elicit tolerance to the disruption of responding maintained on a fixed-ratio schedule of food reinforcement. Similarly in cats, Gillin et al. demonstrated that DMT did not produce tolerance when administered 7-15 days twice daily or every 2 or 24 hours to its effects on EEG, pupil dilation, coordination, posture, and other physical signs. To the contrary, an increase in sensitivity to repeated injections were observed. However, following administration of higher doses of DMT and more frequent injections, partial tolerance to DMT in rats occurred with dose ranges of 3.2 – 10 mg/kg every 2 hours for 21 days. Cross-tolerance to LSD after tolerance to 3.2 mg/kg DMT was established; however, only slight tolerance to LSD was established following 10 mg/kg DMT.
In humans administered 4 repeated doses of DMT 30 minutes apart, Strassman et al. observed no tolerance to the subjective effects of intravenous DMT as measured by the Hallucinogen Rating Scale. However, tolerance did develop to change in body temperature and other physiological factors. Mild cross-tolerance to DMT was reported in humans made tolerant to LSD. Taken together, these findings suggest that tolerance can develop to the cardiovascular and other peripheral effects of DMT, although little or no tolerance develops to the subjective effects.
Subjective effects of DMT
Because the subjective effects of hallucinogens seem to drive their use rather than effects on the reward/reinforcement areas of the brain, drug discrimination is often used as an animal model for testing the behavioral effects of hallucinogens. A compound can be tested for its ability to “substitute”, that is, produce drug-appropriate responding in test subjects trained to discriminate a psychoactive compound from its vehicle or from other psychoactive compounds. Typically, drug-appropriate responding greater than 80% is considered “full substitution”. Conversely, novel compounds can also be trained as discriminative stimuli if they have psychoactive effects, and known compounds can be tested for substitution or antagonism of the novel compound. Asymmetries in cross-substitution can indicate that the two compounds may have overlapping, but not identical mechanisms of action. Drug discrimination can be useful in investigating potential mechanisms of action of the trained discriminative stimulus by utilizing selective agonists and antagonists to either mimic or block the effects. Subsequent paragraphs will examine discrimination studies assessing potential mechanisms of action of DMT.
DMT produced discriminative stimulus effects similar to those of the classic serotonergic hallucinogens DOM and LSD, as DMT fully substituted in DOM-trained rats and produced full or near-full substitution in LSD-trained rats and pigeons. The effects of DMT seem to be mostly hallucinogen-like, as it produced only 50% drug-appropriate responding in MDMA-trained rats, and produced a maximum of 37% drug-appropriate responding in methamphetamine-trained rats. In rats trained to discriminate between the 5-HT2A antagonist ketanserin and the 5-HT2A agonist DOI, DMT produced DOI lever-responding 80% or more of time, indicating that DMT acted more like a 5-HT2A agonist than a 5-HT2A antagonist.
Despite its very short duration of action, DMT can be trained as a discriminative stimulus. A wide range of synthetic phenethylamine hallucinogens fully substitute for DMT, including DOM, DOC, LSD, 2C-D, 2C-E and 2C-I, whereas 2C-C and 2C-T-2 produced a maximum of only 75% DMT-appropriate responding. In contrast, other tryptamine hallucinogens produced more equivocal effects, with DiPT and 5-MeO-DET producing full substitution, 4-OH-DiPT and 5-MeO-IMPT producing partial substitution, and 5-MeO-αMT producing little if any DMT-like effects. In addition, although DiPT fully substituted in DMT-trained rats, DMT only produced 65 % DAR in DiPT-trained rats. Taken together, these findings indicate that serotonergic hallucinogens largely produce discriminative stimulus effects similar, but not entirely identical to those of DMT.
Pharmacological mechanisms
The mechanisms of action for hallucinogens are currently not well understood. The 5-HT2A receptor is thought to be necessary, but not sufficient for hallucinogenic effects, and 5-HT2C and 5-HT1A receptors may play important roles as well. DMT interacts with a variety of serotonin receptors, but also with ionotropic and metabotropic glutamate receptors, dopamine, acetylcholine, TAAR, and sigma-1 receptors.
Serotonin
Most studies to date focus on DMT (and most classic psychedelics) as a partial agonist of serotonin (5-HT) receptors, primarily the 1A, 2A, and 2C receptor subtypes, with predominant interest at 5-HT2A receptors. DMT binds 5-HT1A, 5-HT1B, 5-HT1D, 5-HT2A, 5-HT2B, 5-HT2C, 5-HT5A, 5-HT6 and 5-HT7 receptors with affinities ranging from 39 nM to 2.1 μM. The 5-HT2A receptor is thought to be the primary target of classic serotonergic-mediated psychedelic compounds, such as LSD, DOI, psilocin, and mescaline, although 5-HT1A and 5-HT2C receptors may also play some role. DMT has been reported to bind to all three of these receptors in a variety of studies, including the 5-HT1A, 5-HT2A, and 5-HT2C receptors.
Agonistic properties and affinities for 5-HT1A receptor vary among the classic psychedelics. Interestingly, agonist activity at the 5-HT1A receptor opposes the subjective effect of 5-HT2AR agonists. DMT's affinity for the 5-HT1A receptor is higher compared to 5-MeO-DMT, 6.5 +/- 1.5 nM and 170 +/- 35 nM, respectively. A 5-HT1A antagonist significantly increased the reported psychological effects of DMT. DMT, like other tryptamine hallucinogens, but not phenethylamines, inhibits dorsal raphe cell firing. This mechanism is hypothesized to be an underlying basis of psychedelic-like effects, which may be mediated by stimulation of 5HT1A somatodendritic receptors.
DMT does bind to 5-HT1D receptor and 5-HT3 receptor, however little has been investigated to follow these results up. Delgado shows that 5-HT1 and 5-HT3 receptors exert anxiolytic effects, which does correspond to some reports of DMT use. DMT is an agonist at 5-HT2C receptors. In drug discrimination, the DMT-like effects were partially blocked by a selective 5-HT2C antagonist, SB242084. The 5-HT2C receptor is likely less significant in the psychedelic effects since tolerance develops to the 5-HT2C receptor. Little or no tolerance is developed to the subjective effects of DMT in clinical studies.
DMT binds to the 5-HT2A receptor with relative high affinity, yet other psychedelics that lack visual effects have a higher affinity for the 5-HT2A receptor. The 5-HT2A receptor seems to be necessary, but is not sufficient to account for the visual phenomenon common of the classic hallucinogens. Psychedelics and psychedelic-like compounds including MDMA, 5-MeO-DMT, DET, and DiPT are 5-HT2A receptor agonists. Subjective effects for these compounds are reported to be solely emotional, devoid of visual phenomenon common in other psychedelics such as DMT, except in rare circumstances where individual differences in biology seem to be the regulating factor.
Head twitch response in rodents is thought to be a 5-HT2A receptor-mediated behavior produced primarily by psychedelics, although it is likely that other receptors play a role in this behavior, including 5-HT2C and glutamatergic receptors. Like other classic psychedelics, DMT does induce this head twitch response in C57Bl/6 mice, which is blocked by 5-HT2A inverse agonist, MDL100907. However, the overall number of head twitches induced by DMT is much smaller compared to most other psychedelic compounds. DMT failed to produce this head twitch response in Swiss Webster mice. These discrepancies may be due to the rapid degradation of DMT or other peculiarities specific to DMT.
Functional selectivity on how psychedelic compounds modulate the 5-HT2 receptor family is not well understood. The 5-HT2 family of receptors are Gq/11 mediated and primarily use the phospholipase C second messenger system pathway, but also an phospholipase A2. Phospholipase C hydrolyzes phosphatidylinositol membrane lipids generating inositol-1,4,5-triphosphate (IP3) and diacylglycerate. Diacylglycerate remains bound to the membrane and leads downstream activation of protein kinase C and increases the release of calcium from intracellular stores. In particular, protein kinase C can mediate desensitization of 5-HT2A receptors during drug exposure. Phospholipase A2 stimulation can lead to formation of arachidonic acid. DMT stimulates arachidonic acid release, and less inositol phosphate formation via the 5-HT2A receptor. Whereas inositol phosphate formation via the 5-HT2C receptor seems to be more efficacious and more potent. Stimulation of phospholipase A2 does not seem to directly related to the subjective effects of psychedelic compounds. Other pathways such as the phospholipase D may play a role, but DMT-mediated effects had not been thoroughly investigated. The importance of each second messenger pathway is an important area of future investigation.
DMT, like other classic hallucinogens increase 5-HT levels and/or decrease the turnover of 5-HT. DMT increases excretion of IAA and 5-hydroxy IAA in humans. Other studies have reported an increase in 5-HT and a decrease in 5-hydroxy IAA after DMT administration. DMT seems to have no effect on tryptophan hydroxylase, but produces a main effect on the rate of 5-HT turnover. DMT inhibited SERT transport and VMAT2, acting as a substrate and not as an uptake blocker.
Glutamate and 5-HT/glutamate interactions
An approach gaining increasing interest within the last decade is to examine interacting roles of serotonin and glutamate in mediating the effects of DMT. Of particular interest are the roles of group II metabotropic glutamate receptors, the NMDA receptor, and 5-HT2A receptors in modulating the levels of glutamate in the synapse. These group II glutamate receptors may also be potential target sites for mediating hallucinogenic effects.
mGlu2/3 receptor agonists can act presynaptically to suppress glutamate release, although the significance of this effect in mediating the effects of DMT has not been systematically studied. In contrast, mGluR2/3 antagonist increases the amount of glutamate in the synapse, creating a potentiation of hallucinogenic or psychedelic effects. The 5-HT2A receptor inverse agonist, MDL100907, fully blocked the discriminative stimulus effects and head twitched produced by DMT, whereas the 5-HT2C receptor antagonist produced little or no effect on the discriminative stimulus effects of DMT. A mGlu2/3 receptor agonist produced modest decreases in the discriminative stimulus effects of DMT, whereas a mGlu2/3 receptor antagonist facilitated the effects of low doses of DMT. Comparatively, a mGluR2 agonist blocked the discriminative stimulus effects of LSD, whereas a mGluR2 antagonist facilitated the discriminative stimulus effects of LSD. Further, mGluR2 knockout mice showed little or no head twitch following DOI, and some signaling was disrupted, which may mean that mGlu2 receptors are necessary for hallucinogenic activity. Systematic administration of DOI increases glutamate efflux in ventral tegmental area.
Electrophysiological studies suggest that stimulation of 5HT2A receptors in the medial prefrontal cortex increases pyramidal cell activity and may stimulate corticotegmental glutamatergic projection neurons. A possible explanation for these effects is that mGlu2 receptors co-localize with 5-HT2A receptors to form heteroreceptor complexes. It has been suggested that the heteroreceptors induce a psychedelic-specific second messenger cascade, although this has not been definitively established.
There has been some evidence that NMDA receptors may also play a role in mediating the effects of DMT. DMT partially blocked the discriminative stimulus effects of phencyclidine, which produces hallucinations through its actions at NMDA receptors. In addition, activation of sigma-1 receptor by DMT may lead to potentiation of NMDA receptors.
Dopamine
DMT lacks direct dopaminergic properties, since it did not stimulate dopamine (DA)-sensitive adenylate cyclase. This finding is in agreement with data from a behavioral technique often used to assess direct dopamine agonist effects, which records turning behavior in unilateral nigro-striatal lesioned rats. If a compound stimulates dopamine receptors directly, the animal will rotate toward the intact side, otherwise if a compound induces dopamine release in the striatum from the nerve terminals of the intact side induces a rotation toward the lesion side. As reviewed by Barker, Pieri et al. suggest that DMT appears to have no dopamine receptor agonist effects, although higher doses of DMT does produce ipsilateral turning, although were not indicative of a very potent dopamine releasing effect. Another indication that DMT does not act directly at dopamine receptors is the lack of adenylate cyclase activity in the dorsal striatum of rats.
DMT-induced EEG activation in rabbits can be antagonized by neuroleptics (DA receptor blocking compounds. This may be due to blockade of downstream sequela. For example, DMT releases dopamine from presynaptic stores. This release of dopamine in combination with the effects of MAO causes an indirect dopaminergic stimulant activity. DMT caused 42% decrease in concentration of dopamine in rat forebrain, while norepinephrine was not affected, no change in the levels of the dopamine metabolite homovanillic acid was observed in corpus striatum. This decrease in concentration of dopamine may be caused by a stimulation of the release of dopamine or by inhibition of its synthesis. This decrease in dopamine levels is likely not related to change in synthesis, because no change in norepinephrine levels or turnover rate in the diencephalon were observed. It appears as if DMT increases central dopamine turnover and enhances striatal dopamine synthesis in rats.
Acute and chronic administration of DMT significantly increased endogenous levels of striatal 3-MT. Dopamine steady state concentrations remained unchanged. Further, DMT increased accumulation of 3HDA and 3H3MT newly formed from 3HDOPA. DOPAC, a major metabolite of dopamine more efficiently lowered by DMT rather than HVA in the striatum and whole brain. This is distinct from the effects of classic MAOIs, which decrease both DOPAC and HVA. After acute administration striatal dopamine synthesis was increased, yet there was no effect on steady state conditions. Dopamine degradation must be enhanced proportionally and is likely done so extraneuronally, due to the increase in 3-MT. No change in the increase of DA turnover over one month treatment, with consistent rises in 3-MT is observed.
Acetylcholine
Little investigation has occurred in reference to DMT's effect on acetylcholine. DMT significantly decreases concentration of acetylcholine in corpus striatum, which may be due to a direct release of acetylcholine, thus reducing concentration of striatal acetylcholine. Generally, acetylcholine levels in brain are reduced when its rate of release or turnover are increased. DMT had no effect on the level of acetylcholine in the cortex.
Trace amine-associated receptors
Trace amine-associated receptors (TAARs) are a more recently discovered class of receptors which may play a role in mediating DMT and other psychedelic drug effects. The rat trace amine-associated receptor – 1 (rTAAR1) is a G protein-coupled receptor with homology with members of the catecholamine receptor family. Trace amines p-tyramine, and p-PEA stimulate cAMP production, are commonly measured to assess activation of the TAAR. DMT binds to the rTAAR-1 with high affinity and acts as an agonist, causing activation of adenylyl cyclase and resultant cAMP accumulation in HEK293 cells transfected with rTAAR1. Other psychedelics such as (+/-)DOI, d-LSD, and 5-MeO-DMT, and non-psychedelics such as R(+)lisuride, (+/-)MDMA and amphetamine also stimulate cAMP production through their effects at rTAAR1. This second-messenger cascade does not seem to be selective for any of these compounds as these effects occurred at approximately 1 μM concentration (no other concentrations tested). rTAAR1 seems to be located in the intracellular puncta, and not at the plasma membrane in vitro; it is not known if this is the case in vivo.
Because TAARs were discovered long after research had on DMT (and other psychedelic compounds) had been initiated at the 5HT2AR, there is a paucity of research on the role of TAAR, which makes it difficult to discern what role this class of receptor may play in mediating the effects of endogenous and exogenously administered DMT. It is unknown whether the typically used 5-HT2AR antagonists ketanserin and/or risperidone have any antagonist effects of TAAR as well. This is an area where more research needs to be done to fully understand the importance of TAARs and psychedelic effects.
Sigma-1 receptor
The sigma-1 receptor was once thought to be a subtype of an opioid receptor. It has been implicated to have a role in several neurobiological diseases and conditions such as addiction, depression, amnesia, pain, stroke, and cancer. It is found widely distributed though out the body including in the CNS, liver, heart, lung, adrenal gland, spleen, and pancreas. They are localized between endoplasmic reticulum and mitochondrion. Sigma-1 receptor agonists signal the receptor to disassociate itself form other endoplasmic reticulum chaperones, which allows the receptor to act as a molecule chaperone to IP3 receptors. This enhances calcium signaling from the ER to mitochondria, activates TCA cycle and increase ATP production. Sigma-1 receptors can translocate to plasma membrane or sub plasma membrane area when stimulated with higher concentrations of agonists or when sigma-1 receptors are over-expressed. Once sigma-1 receptors translocate to the plasma membrane they can interact with and inhibit several ion channels. Sigma-1 receptor activation can also lead to potentiation of NMDA receptors.
Psychedelics and non-psychedelics bind promiscuously to sigma-1 receptors. DMT binds to sigma-1 receptors at low micromolar concentrations, and appears to have agonist-like effects. DMT inhibits cardiac voltage-activated sodium ion channels at higher concentration (100 M) in HEK293 cells, and neonatal mouse cardiac myocytes, induces hypermobility in wild-type mice, which is blocked in sigma-1 receptor knock-out mice. DMT modulated current in sigma-receptor-mediated Na+ channels, which was reduced by sigma-1 receptor knockdown and by progesterone. In addition, DMT synthesizing enzyme indolethylamine-N-methyltransferase is co-localized with sigma-1 receptor in C-terminals of motor neurons, which suggests that there may be adequate levels of endogenous DMT to activate sigma-1 receptors.
The main problem with the theory that DMT is an endogenous sigma-1 receptor agonist is that it requires concentrations in the micromolar range, whereas selective sigma-1R agonists such as (+)-pentazocine have affinities in the nanomolar range. If DMT is only available in trace amount in humans and is rapidly metabolized, how can DMT levels rise enough to account for sigma-1 receptor-mediated effects? One possible explanation for this is the three step process of accumulation and storage discussed earlier, which includes active transport across the blood brain barrier, and DMT may be a substrate for transporters at the cell surface and at the neuron level. Supporting the role of sigma-1 receptor is that the SSRI fluvoxamine, has sigma-1 receptor agonist properties with higher affinity than DMT. Fluvoxamine works better with patients suffering from psychotic depression compared to antidepressants without sigma-1 receptor agonist properties. Selective sigma-1 receptor agonists do not cause psychotomimetic effects in animals. At best, sigma-1 receptors may partially mediate the subjective effects of DMT.
Whether or not the sigma-1 receptor plays a significant role in the psychedelic effects of DMT, it may still play an important role in other physiological mechanisms. Sigma-1 receptors agonists are potentially neuroprotective via several mechanisms. DMT reduced inflammation ostensibly via sigma-1 receptor, and can induce neuronal plasticity, which is a long-term recuperative process that goes beyond neuroprotection. Sigma-1 receptors can regulate cell survival and proliferation, thus if DMT is an endogenous agonist, this may explain physiological relevance and importance of why DMT has 3-step uptake process.
Regulation of intracellular calcium overload, proapoptotic gene expression via Sigma-1 receptors, can result in neuroprotection during and after ischemia and acidosis. There would be further benefit through sigma-1 receptor dependent plasticity changes. Along these lines Frecska colleagues suggest that DMT may be protective during cardiac arrest, beneficial during perinatal development, immunoregulation, and aid in reducing cancer progression.
Immediate early gene stimulation
Through second messenger systems, DMT can affect the rate of genetic transcription, such that DMT encodes the transcription factors c-fos, egr-1 and egr-2, which are associated with synaptic plasticity. Increases in expression of brain-derived neurotrophic factor (BDNF) are also observed after DMT administration. BDNF expression is associated with synaptic plasticity, cognitive process such as memory and attention, and modulation of efficacy and plasticity of synapses.
Summary
As previously mentioned, DMT interacts with a variety of ionotropic and metabotropic receptors. The subjective effects of large doses of exogenous DMT are most likely mediated primarily by 5-HT2A receptors, with 5-HT2C receptors playing little or no role. mGlu2/3 receptors have significant modulatory effects, and the interaction of serotonergic and glutaminergic receptors may play a central role. DMT does not have direct effects on DA receptors, but indirectly alters the levels of dopamine, with resulting neurochemical and behavioral effects. Similarly, DMT also alters levels of acetylcholine. Finally, DMT may be an endogenous ligand at TAAR and sigma-1 receptors, but at the least, the effects of DMT at these receptors may play important physiological roles.
DMT as a model of psychiatric disorders
There has been a revival of interest in clinical uses of hallucinogens. Among the first were a series of controlled clinical studies on DMT. Those studies reported that pure DMT had rapid and extremely strong cardiovascular effects as well as profound psychological effects. The cardiovascular effects preclude the use of pure DMT; however, ayahuasca and other DMT-containing ritual beverages seem to be less toxic while retaining the psychological effects. Based on studies of the health status of ayahuasca users, the use of ayahuasca may be safe and even beneficial.
Recently, a series of studies examined the long-term personal and spiritual significance of exposure to psilocybin have suggested that psilocybin may be useful for anxiety-related disorders. Similarly, ayahuasca and similar DMT-containing mixtures have been proposed as treatments for a variety of psychiatric disorders and ayahuasca is mostly well-tolerated. For example, long-term ayahuasca users showed less psychopathology, and better performance on neuropsychological tests compared to matched controls and less substance abuse and fewer psychiatric/psychosocial problems than matched controls.
Schizophrenia
The classic positive symptoms of schizophrenia include delusions and hallucinogens, so hallucinogenic compounds seem an obvious tool for modeling schizophrenia. Given that hallucinogens produce their effects primarily through activation of the 5-HT2A receptor, the serotonin system provides an alternative to the dopamine model of schizophrenia. The dopamine model has produced a wide range of treatment medications which are very useful, but do not fully treat the range of symptoms experienced during psychotic episodes and produce substantial adverse effects. Discovery that DMT exists as an endogenous compound led to research focusing on DMT as a model of schizophrenia in the 1960s and 1970s. Reviews of this early research concluded that the data was suggestive but not conclusive. These early studies are not reviewed in the present manuscript.
Subsequent research reported that levels of endogenous DMT increased in schizophrenic patients during psychotic episodes, which declined as their state improved. However, no changes in DMT levels were observed in rapidly cycling states (manic-depressive). These findings renewed interest in the transmethylation hypothesis, which states that schizophrenia may be due to stress-induced production of psychotomimetic methylated derivatives of catecholamines or indolealkylamines in the brain. DMT seems to fits the bill as it is an indolealkylamine, is an endogenous compound, and is linked to stress reactivity.
In addition, DMT was identified as the active ingredient in ayahuasca, which produces effects similar to a psychotic episode, including thought disorders, delusions, and hallucinations. When given to human subjects, DMT produces complex visual and auditory hallucinations and increases cortisol levels, which supports its possible role as a possible mediator of schizophrenia.
More recent studies have examined the effects of DMT on various experimental models of changes in cognition in schizophrenic patients. Normal subjects are administered DMT and given various cognitive tasks to perform during fMRI scans. DMT slowed reaction time in tests of inhibition of return, decreased alertness, but produced less mismatch negativity than did the NMDA glutamate channel blocker ketamine, which commonly serves as a tool for investigating the glutaminergic hypothesis of schizophrenia.
In summary, DMT is still an interesting model of the serotonergic aspects of schizophrenia, but there is no conclusive evidence that endogenous DMT is a primary player. In fact, it has been argued that DMT is anti-anxiety/anti-psychotic via actions at the trace amino acid receptor (TAAR). Jacob and Presti, and others have suggested that the effects of endogenous DMT are mediated via sigma receptor roles.
Depression
Few studies have investigated the effects of DMT-containing compounds on depression. One study investigated the effects of ayahuasca in the forced-swim test, a common animal model of depression. In female Wistar rats, ayahuasca increased swimming, which is considered a sign of potential antidepressant effects. In a human experimental study, long-term ayahuasca users showed reduced ratings of hopelessness while under the influence. Finally, in an open-label clinical trial in in-patients suffering from depression, ayahuasca produced marked improvement in depressive symptoms with no mania or hypomania for up to up to 21 days after a single dose. Convergent evidence from three different experimental approaches provides stronger evidence for potential antidepressant effects of DMT. However, replication of these findings will be necessary to confirm whether DMT-containing compounds will be useful for treatment of depression.
Anxiety/aggression
It has been proposed that DMT is an endogenous anxiolytic compound through its actions at the trace amino acid receptor. To date, this hypothesis has generated little interest and DMT has been mostly investigated for its hallucinogenic effects. One early study did examine the effects of DMT in an animal model of anxiety/aggression in which pairs of rats receive shocks while in a test chamber. The shocks produce fighting and anti-anxiety compounds reduce the shock-induced fighting. LSD increased the amount of fighting, whereas DMT suppressed fighting. However, the effective doses also produce sedation and reduced locomotor activity, which could also account for the effects.
In a case study of a homeless male with multiple convictions for manslaughter and diagnosed with antisocial disorder, ayahuasca sessions reportedly produced significant moral insights and allowed completion of a rehabilitation program in which the subject had been highly resistant. No follow up was conducted, so no data is available on whether incidences of violent behavior decreased. In two larger scale studies, ayahuasca decreased ratings of anxiety in depressive-disorder patients and reduced ratings of panic but not state- or trait-anxiety, in long-term users. Taken together, these findings do not provide support that DMT is useful for treatment of anxiety and/or aggression. It is possible that DMT may be useful in specific settings, similar to the successful use of psilocybin to treat anxiety in cancer patients, but careful experimental research will be necessary before a strong conclusion can be make about DMT's efficacy as an anxiolytic medication.
Effects on cardiovascular system
Single doses of DMT produced rapid onset of marked sympathomimetic effects including increased heart rate and blood pressure. When a 5-HT1A antagonist, pindolol, was co-administered with DMT, the increase in heart rate was diminished whereas the increase in blood pressure was enhanced. Tolerance to the effects of DMT was tested by administration of DMT to human volunteers four times at 30-min intervals. A progressive decrease in heart rate was observed over the four doses, but not in blood pressure. In contrast, two repeated doses of ayahuasca 4-h apart reduced systolic blood pressure and heart rate. Long-term use of DMT-containing beverages may be of more concern as 14-day exposure to ayahuasca in rats altered the structure of the aorta, leading to a thickening of the walls of the aorta relative to the lumen diameter.
Cardiac arrest
DMT has been speculated to aid in extending the survival of brain. A review by Frecska and colleagues suggests that during physical signals of agony, lungs synthesize large amount of DMT and can release DMT into arterial blood within seconds. Once in blood circulation DMT is safe from degradation as extracellular, circulating MAO enzymes deaminate only primary amines. DMT is a tertiary amine, thus reaching the brain with minimal degradation. Through the use of active transport mechanisms already discussed for taking DMT from blood into the brain, could potentially keep brain alive longer without the brain having to produce DMT on its own. Exogenous DMT-like psychedelic effects are in essence similar to subjective reports provided after clinical death and near death experiences. Strassman believes DMT to be very likely involved in the dying process.
Endocrine system
DMT increased levels of corticotropin, cortisol, prolactin, and growth hormone when administered to human volunteers. When DMT was given repeatedly to human volunteers, tolerance to the increases in various endocrine levels was observed, including corticotropin, prolactin and cortisol. Similarly, ayahuasca increased prolactin and cortisol levels in human volunteers, whereas repeated doses resulted in lower levels of GH secretion.
Immune system and neurotoxicity
Ayahuasca has been reported to decrease the percentage of CD3 and CD4 lymphocytes, but to increase the number of natural killer cells. It has been hypothesized that DMT might increase activity of the immune system and could prove useful as a treatment for cancer. Evidence for this hypothesis is equivocal. DMT increased the cytotoxic activity of peripheral blood mononuclear cells in the A172 human glioma cell line. However, in another study, DMT did not exhibit cytotoxicity of KB or HepG2 carcinoma cells. In addition, others have proposed that DMT and related compounds are anti-inflammatory and reported that DMT inhibited production of pro-inflammatory compounds IL-1β, IL-6, IL-8 and TNFα and increased levels of the anti-inflammatory compound IL-10 through actions at the sigma-1 receptor.
Immunoregulation
Serotonin plays an important role with immunoregulation. And on cellular immune functions critical in the elimination of pathogens or cancer cells. It is possible that DMT may also play a role in immunoregulation via its Sigma-1 and 5-HT2A receptor activation. Sigma receptors are also expressed on many cells of the immune system. In particular, Dorocq showed that sigma-1 receptors can reduce pro-inflammatory cytokines and enhance the production of anti-inflammatory cytokine IL-10. DMT through the formulation of ayahuasca increased levels of blood circulating natural killer (NK) cells with concentrations as low as 1 mg DMT/kg body weight. In vitro DMT administration has shown an increase of secreted interferons in vitro in NK cell and dendritic cell cultures. Interferons are potent anticancer factors. If DMT does increase interferon secretion, it may be beneficial in contributing to or aid in better elimination of malignant and/or infected cells.
Perinatal INMT activity
Levels of INMT in the placenta are higher than in adults. It is speculated that activity in fetal lungs compensates for difference. INMT activity in rabbit lung is relatively high in fetus, increases rapidly after birth and peaks at 15 days of age. It then declines to mature levels and remains constant through life. If INMT levels are paralleled with increased DMT synthesis, it could be possible that DMT-mediated sigma-1 receptor activity induces neuronal plasticity changes that can be expected for newborns. Selective sigma-1 receptor agonists have shown to be protective against excitotoxic perinatal brain injury and ischemic neurodegeneration in neonatal striatum. Expression of INMT seems to be important for pregnancy success. Whether DMT, a product of INMT plays any role in these protective and beneficial effects, is unknown.
INMT and cancer
Down regulation of the expression for the gene responsible for INMT production has been associated with cancer. It is believe to be a potential candidate gene in prevention of cancer progression. INMT expression has been associated with a dramatic decrease in recurrence of malignant prostate and lung cancers. It is possible that the regulating roles of INMT via its product DMT could potentially have a direct tumor suppression effect, but this is highly speculative.
Summary and conclusions
DMT is a compound found widely across the plant and animal kingdoms. In mammals, the psychoactive effects produced by DMT seem to be largely mediated by the 5-HT2AR, although the complex subjective effects reported by DMT users are likely modulated by other receptor systems such as the metabotropic glutamate receptors.
The wide use of DMT in the form of ayahuasca for many years has led to a number of studies focusing on adverse health effects or potential benefits of ayahuasca use. There have been few reports of adverse health consequences. Ayahuasca did produce modest impairment of cognitive function in inexperienced users; however, little or no impairment was observed in experienced users. Ayahuasca decreased markers of sleep quality and sleep disturbances are common on the night following administration, but the users reported no perception of deterioration of quality. As mentioned previously, there is little sign of tolerance or dependence to DMT except to the cardiovascular and endocrine effects, which actually could be viewed as the primary adverse effects. Diminution of these effects would preferred by long-term users. The greatest concern appears to the possibility of teratogenicity. Large doses of ayahuasca 50-fold higher than typical ritual doses were fed to pregnant rats. No lethality was observed, but increased incidence of cleft palate and skeletal malformations was observed in their pups.
DMT may be an agent of significant adaptive mechanisms that can also serve as a promising tool in the development of future medical therapies. There have been proposals that DMT might be a useful treatment of anxiety, substance abuse, inflammation, or for cancer. Experimental studies have been few and it is premature to conclude that DMT may have clinically relevant uses.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5048497/