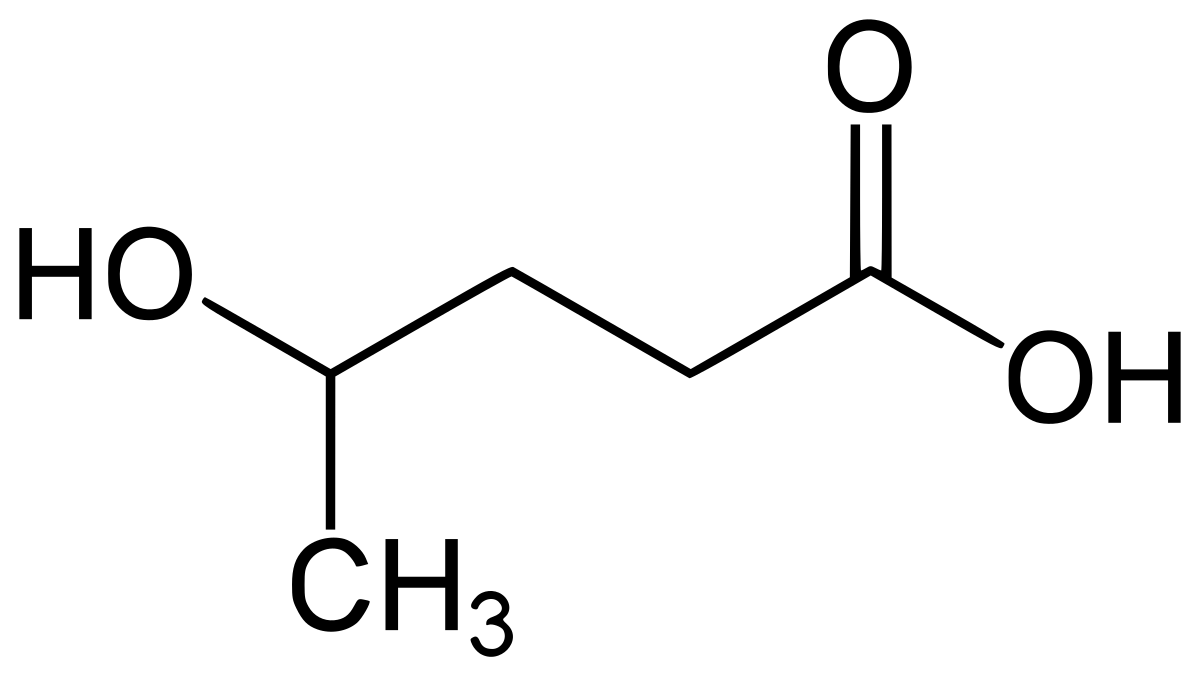
As many of us know, the majority of Pregabalin (Lyrica) is excreted unchanged, but a very small portion is metabolized into 4-Methylpregabalin.
This is false. A very minor portion of pregabalin is metabolized into N-methyl pregabalin, not 4-methyl-pregabalin.
I'm very curious though. There are some other Gabapentinoids that are allegedly under development that I'm sure are going to be quite interesting. The patent on Lyrica finally ran out this year so you can only imagine they're scrambling to raise another prize pig as fast as they can. I've never used GHB

Weirdly enough, the search for a successor to pregabalin hasn't been all that fruitful for Pfizer.
While they were quick to discover a number of analogues that had a much higher affinity for the target ion channels
in-vitro, this didn't translate into increased
in-vivo potency. This is because the drug is too polar to enter the brain on its own, so it has to rely on certain transporter proteins, which are normally responsible for the uptake of the amino acid leucine. This naturally complicates the development of such a drug, since it has to be designed to fit into not one, but two different pharmacophores.
A few drug candidates (atagabalin, imagabalin, 4-methylpregabalin) apparently made it into clinical trials, but it looks like Pfizer eventually shelved them all after failing to demonstrate any additional therapeutic benefits when compared to pregabalin itself. Mirogabalin (which was developed by Daiichi-Sankyo, not Pfizer) recently got approved for neuropathic pain in Japan, but as far as I've read that's it for novel gabapentinoids.