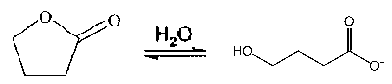No problem

My experience with GHB is very limited, so that's only what I read and heard: GBL gets absorbed more rapidly than GHB due to it's more lipophilic nature. Na-GHB gets protonated in the stomach, so in this state it's able to cross the lipid barriers of the membranes. Due to chemical equilibrium there's a part of the GHB that stays ionic and can't get absorbed because it's too polar. This means a part of the GHB still sits around in the stomach, unable gettin' absorbed, what prolongues the onset and the overall duration.
This reaction shows the hydrolysis of GBL to GHB, one can see that GBL is a direct precursor/prodrug:
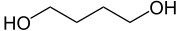
If you compare GHB to 1,4-Butanediol it get's clear that there's more work to do to get to the desired GHB:
One of the carbon next to the alcohol groups (-OH) needs to be oxidized all the way up to an acid. But first comes the aldehyde, which is propably not really enjoyable. All aliphatic alcohols get metabolized the same way: Alcohol (-OH) -> Aldehyde (-CHO) -> Carboxylic acid (-COOH), because the latter is the most water soluble and can thus get rid of in urine.
When our example is ethanol, the first metabolite is ethanal/acetaldehyde, which is quite nasty and causes the notorious hangover that's associated with ethanol consumption.
So it's at least not unlikely for the aldehyde intermediate in the GHB biosynthesis to behave similar.
But you see, there's two alcohol groups, that means both of them can be oxidized to an aldehyde, and again, it wouldn't surprise me if this possible metabolite is toxic, too.
Long story short: While GHB is definetly a metabolite of 1,4-Butanediol, there are several other metabolites (not mentioned all possible ones), that can and will be produced, hence the higher toxicity and "dirtier" high in comparison to pure GHB