-
Select Your Topic Then Scroll Down
Alcohol Bupe Benzos Cocaine Heroin Opioids RCs Stimulants Misc Harm Reduction All Topics Gabapentinoids Tired of your habit? Struggling to cope?
Want to regain control or get sober?
Visit our Recovery Support Forums -
OD Moderators: Keif’ Richards | negrogesic
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
RCs Novel stimulant/empathogen Mexedrone
- Thread starter roi
- Start date
bjznoviskey
Bluelighter
- Joined
- Oct 17, 2013
- Messages
- 119
I think an n-acetyl might have been a better choice, I would consider putting that in my body. This, I don't think so. It'll probably do "something" but will it live up to the hype, unlikely
Shulgin's attempt at n-methoxy
"The ultimate reason for making MDMEO was, of course, that it could be made. That reason is totally sufficient all by itself."
Shulgin's attempt at n-methoxy
"The ultimate reason for making MDMEO was, of course, that it could be made. That reason is totally sufficient all by itself."
Last edited:
Rybee
Bluelighter
- Joined
- May 29, 2013
- Messages
- 1,305
Heard of a lot of he-say-she-say about this substance so would be very interested to hear of any first account trip reports on this one please.
There's also been a lot of debate about whether this will be legal in the UK, I know the rules on legal advice/discussion, but several chemical names have been touted. Is anybody able to 100% confirm its' structure so that I can determine its' legality for myself?
There's also been a lot of debate about whether this will be legal in the UK, I know the rules on legal advice/discussion, but several chemical names have been touted. Is anybody able to 100% confirm its' structure so that I can determine its' legality for myself?
adder
Bluelighter
- Joined
- Mar 28, 2006
- Messages
- 2,851
Heard of a lot of he-say-she-say about this substance so would be very interested to hear of any first account trip reports on this one please.
There's also been a lot of debate about whether this will be legal in the UK, I know the rules on legal advice/discussion, but several chemical names have been touted. Is anybody able to 100% confirm its' structure so that I can determine its' legality for myself?
I don't think anyone can confirm the structure for you now. It could be the compound from the first post which, like I wrote, perfectly matches reports on normephedrone. 4-methyl-N-methoxycathinone or N-methoxynormephedrone is likely metabolised to normephedrone in vivo and reports of a less stimulating, more sedating, MDAI-like substance would confirm it if that's what it really is. I don't know the analogue laws in the UK but it's possible that they don't mention hydroxy and methoxy substituents on the amine nitrogen. The similar situation was encountered with the HOT series of psychedelics which were perfectly legal in some places where their 2C-T counterparts, which are likely produced in vivo from HOT compounds, were not.
Mind you that normephedrone never made it to the wide public during the time when first mephedrone replacements like 3-MMC were available. Don't expect anything mind-blowing from this fuss on mexedrone, it's not what it was painted to be, there were at least a few brilliant compounds that I read about here on BL and they're gone for good without even being widely released. If it was that good, I'm sure it would have flooded the clubs before anyone could even know the structure. Besides less and less people are still naive enough to believe the RC business could be any different from other businesses generating huge revenue with relatively low expenditure. Not that it's going to change marketing techniques but perhaps can work well for the quality of RCs in the future. I mean, after all the society recognises the need for a narcotic like alcohol to exist even though there are no less downsides to it than to many illegal psychoactive compounds, so what's wrong with careful design of synthetic drugs? However, I'm not sure if more of the unknown is what we really need now in this field when there are many illegal compounds still waiting to be researched.
Last edited:
doppelgänger1
Bluelighter
- Joined
- Feb 24, 2012
- Messages
- 160
Would the n-methyl,n-methoxy-analogue then yield 4-MMC?
roi
Bluelighter
- Joined
- Sep 2, 2013
- Messages
- 1,545
We might have gotten bamboozled, if WEDINOS is right.
http://www.wedinos.org/db/materials/view/00716
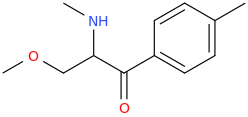
Methyl ether of Mephedrone? I'll update the first post.
http://www.wedinos.org/db/materials/view/00716

Methyl ether of Mephedrone? I'll update the first post.
Last edited:
how should that be legal in the UK ?
"3. Any ester or ether of a substance specified in paragraph 1 or 2."
(i don't believe the story that was presented on BL that you can speak of "ether of" only if the parent molecule has a hydroxy on the position where the ether gets attached)
"3. Any ester or ether of a substance specified in paragraph 1 or 2."
(i don't believe the story that was presented on BL that you can speak of "ether of" only if the parent molecule has a hydroxy on the position where the ether gets attached)
roi
Bluelighter
- Joined
- Sep 2, 2013
- Messages
- 1,545
In the UK class A and C esters and ethers are illegal, but for unknown reasons not Class B. So this one is legal in the UK.
Cathinones are class B.
psynce of sound
Bluelighter
- Joined
- Aug 14, 2014
- Messages
- 386
I have procured a gram sample of this seeming conundrum. Purely out of curiosity and acquisition of data.
Any information of dosage (anecdotal or research derived)
I have consumed 145mg (admittedly with 3-FPM).
I found that it was very very sedating for the first hour or so. So much I added a 30mg plug of more 3F to combat sleep and to see if I can achieve desired outcomes.
All in it was a sutble experience bar the sedation early on. I'm going to try solo trials soon. Would like to know more about dose even if purely speculative.
Will give some ground to then create logical assertions.
Peace guys.
Any information of dosage (anecdotal or research derived)
I have consumed 145mg (admittedly with 3-FPM).
I found that it was very very sedating for the first hour or so. So much I added a 30mg plug of more 3F to combat sleep and to see if I can achieve desired outcomes.
All in it was a sutble experience bar the sedation early on. I'm going to try solo trials soon. Would like to know more about dose even if purely speculative.
Will give some ground to then create logical assertions.
Peace guys.
zenit992
Greenlighter
The he-say-she-say about this is that it is utter crap. There is a thread about this compound in European and African Drug discussions: http://www.bluelight.org/vb/threads/764088-Mexedrone
perhaps they should be merged?
perhaps they should be merged?
adder
Bluelighter
- Joined
- Mar 28, 2006
- Messages
- 2,851
adder,
I didn't name / draw N-methoxy-nor-mephedrone. Mexedrone is, purportedely, alpha-methoxymethyl-mephedrone. The only "N-" in this case methyl.
The structure in the first post was changed after my last post in the thread. It was the structure of N-methoxynormephedrone which mexedrone was rumoured to be. I don't know if it really is. The new structure is not a methyl ether of mephedrone, perhaps beta-methoxymephedrone if you count C atoms from the carbonyl group, anyway, I doubt there are any sources that could justify the choice of such a substitution while you could certainly find some sense in N-MeO-normephedrone in theory.
zenit992
Greenlighter
This thread at least has the structure in the first post so people aren't totally confused.
I wish there was a proper RC subforum.
True, but when that thread was started no one knew the structure; it was kept secret up until release for some reason.
I agree, an RC subforum would be a good ideea.
adder
Bluelighter
- Joined
- Mar 28, 2006
- Messages
- 2,851
From what I've seen, I guess both compounds might be in circulation right now, mexedrone appears to be beta-methoxymephedrone indeed (the structure in the first post).
I've read some weird posts on one Polish forum where some people state that mexedrone (beta-methoxymephedrone) is very weak, almost inactive, however, upon contact with air for a few hours it magically becomes much more potent. Very weird I thought. But then I started wondering if slow condensation with the removal of CH3OH could be possible, the resulting product would be an enone, also unstable under humid conditions due to the presence of amine (basically methylamine with a substituted vinyl group), but perhaps stable enough to be ingested. It would be very similar in structure to mephedrone itself but with an alpha-beta double bond.
I've read some weird posts on one Polish forum where some people state that mexedrone (beta-methoxymephedrone) is very weak, almost inactive, however, upon contact with air for a few hours it magically becomes much more potent. Very weird I thought. But then I started wondering if slow condensation with the removal of CH3OH could be possible, the resulting product would be an enone, also unstable under humid conditions due to the presence of amine (basically methylamine with a substituted vinyl group), but perhaps stable enough to be ingested. It would be very similar in structure to mephedrone itself but with an alpha-beta double bond.
Last edited:
roi
Bluelighter
- Joined
- Sep 2, 2013
- Messages
- 1,545
The N-methoxy-4-MC was mentioned on a polish blog iirc, kind of a shitty bait.
I'm pretty sure that all Mexedrone right now available is the beta-methylmethoxy-mephedrone.
I'd love to see N-Hydroxy-4-MMC (as seen in MDOH). Not UK legal but the rest of the world would love it.
I'm pretty sure that all Mexedrone right now available is the beta-methylmethoxy-mephedrone.
I'd love to see N-Hydroxy-4-MMC (as seen in MDOH). Not UK legal but the rest of the world would love it.




