Image 1 hosted in ImgBB

ibb.co
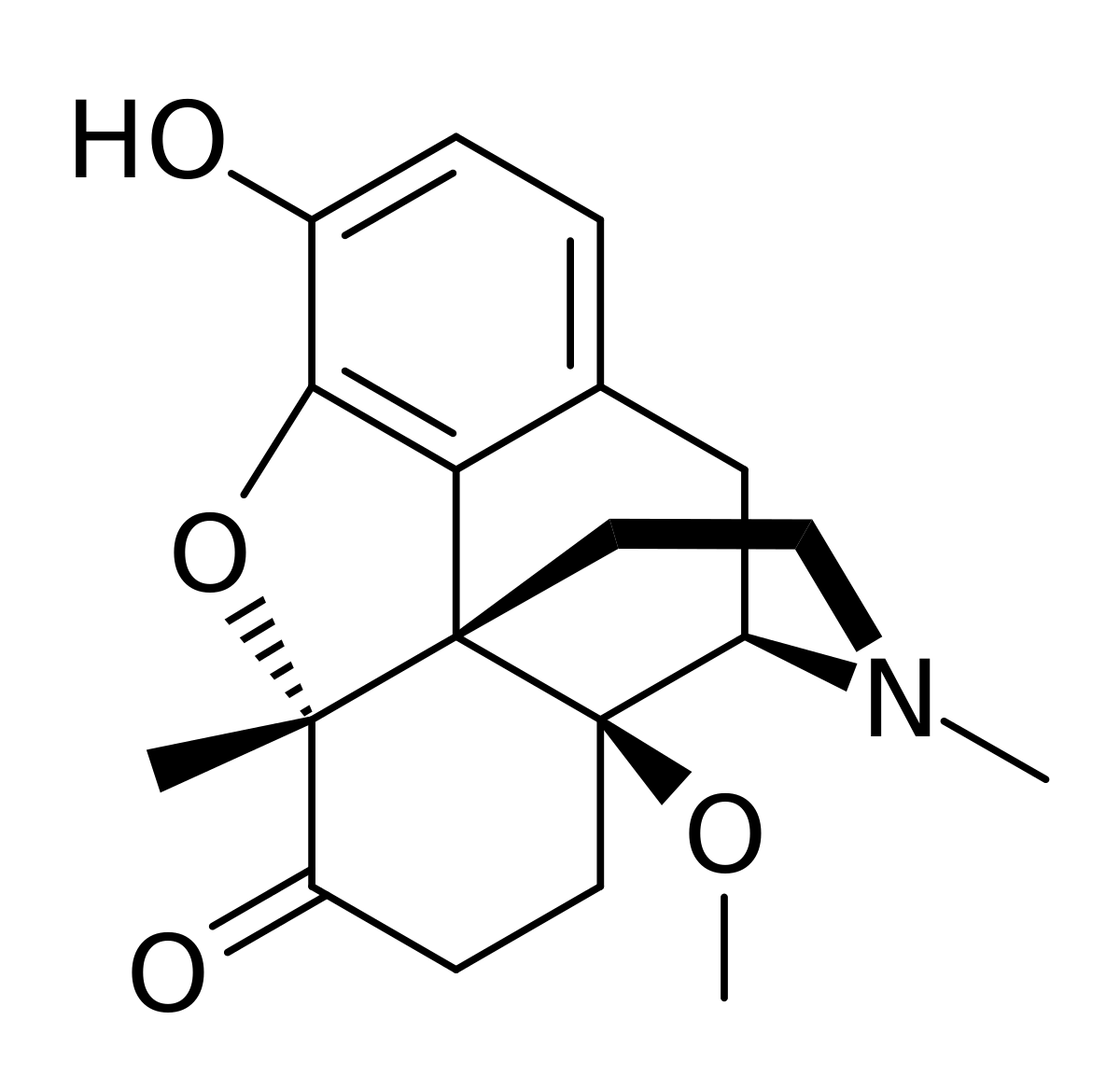
The only modification medicinal chemists might not immediately understand is the replacement of a 3-hydroxy for a 3-carboxamide. This actually halves the potency BUT does substantially increase duration of action by removing the major metabolic pathway of 3-gluconation as demostrated:

en.wikipedia.org
As for the other modifications. The (S)-beta hydroxy N-2-(2-furyl)ethyl moety:
The 14-methoxy:

en.wikipedia.org
The 6-methylene:

en.wikipedia.org
In essence, QSAR analysis suggests that the potency of the compound will certainly be in the range of thousands of times that of morphine BUT importantly, all of the references suggest a hugely increased TI. While I would be VERY careful in exploring this aspect, it would appear that while much more potent than morphine, the lethal dose due to respiratory collapse will be similar to that of morphine.
Of course the downside is that the affinity would mean that naloxone is of limited use in the case of overdose. That said, it would appear to be of limited utility in cases of overdose with fentanyl and carfentanil. That said, M5050 has been known since the early 1970s and whenever etorphine or carfentanil as used (only for large mammals), protocol dictates that M5050 is also available in case of accidental ingestion:

en.wikipedia.org
My limited knowledge of diprenorphine is that it's restricted use is due to it's side-effects, specifically hallucinations and dysphoria. I've never quite understood the resistance to the use of diprenorphine when fentanyl or carfentanil overdosage is possible beyond the fact that it's quite an expensive drug to manufacture and since all patents have expired, their is no obvious profit-motive for producers.
I do know that KW bentley and his team also developed the N-allyl homologue of diprenorphine which I presume will have a significantly shorter duration of action but it's interesting that even by the 1960s the N-methylcyclopropyl moiety was favoured over the N-allyl moiety.