-
N&PD Moderators: Skorpio | thegreenhand
-
Neuroscience & Pharmacology Discussion Welcome Guest
Posting Rules Bluelight Rules Recent Journal Articles Chemistry Mega-Thread FREE Chemistry Databases! Self-Education Guide
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Rectify's molecular poetry thread
- Thread starter Dresden
- Start date
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
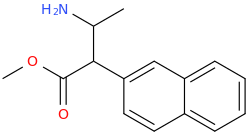
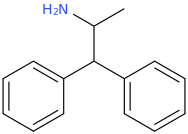
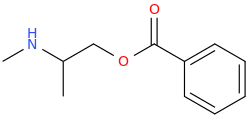
ASHTON
1-phenyl-1-carbophenoxy-2-methylaminopropane
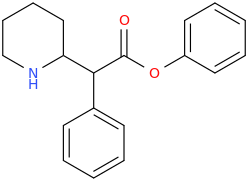
RIHANNA
1-phenyl-1-carbophenoxy-1-(2-piperidinyl)-methane
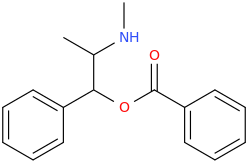
COCA_PSEUDA
1-phenyl-1-(1-oxa-2-oxo-2-phenylethyl)-2-methylaminopropane
from ephedrine and sodium phenylcarboxylate
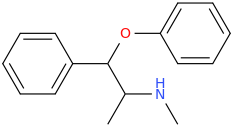
AUSTIN
1-phenyl-1-phenoxy-2-methylaminopropane
from ephedrine and sodium phenoxide
BOLIVIAN_MARCHING_POWDER
2-methylamino-3-(1-oxa-2-oxo-2-phenylethyl)-propane
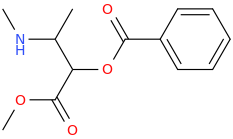
PERUVIAN_FLAKE
2-methylamino-3-(1-oxa-2-oxo-2-phenylethyl)-3-carbomethoxy-propane
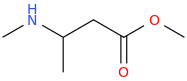
FISHSCALE
2-methylamino-3-carbomethoxy-propane
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
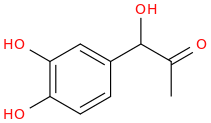
1-(3,4-dihydroxyphenyl)-1-hydroxy-2-oxopropane
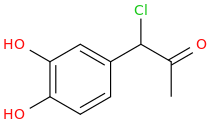
1-(3,4-dihydroxyphenyl)-1-chloro-2-oxopropane

1-(3,4-dihydroxyphenyl)-1-cyano-2-oxopropane
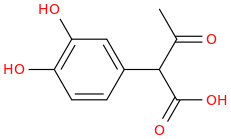
1-(3,4-dihydroxyphenyl)-1-carboxy-2-oxopropane
CH2Cl2 / KOH
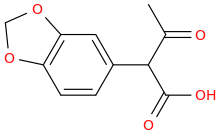
1-(3,4-methylenedioxyphenyl)-1-carboxy-2-oxopropane
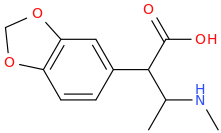
1-(3,4-methylenedioxyphenyl)-1-carboxy-2-methylamino-propane
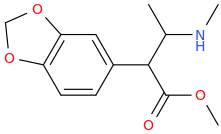
THE_L.O.V.E_DRUG in 6 Steps
1-(3,4-methylenedioxyphenyl)-1-carbomethoxy-2-methylamino-propane
This Synthetic Outline Cleverly Avoids The Harsh Reaction Conditions (either H+ or -OH with heat in reflux) Which Could Destroy The Acetal But Which Are Required For The Reaction Of An R-Cyano Functional Group To An R-CO2H Functional Group.
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
Fun With Benzyl Piperazines!
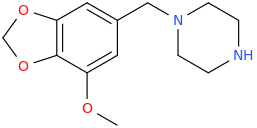
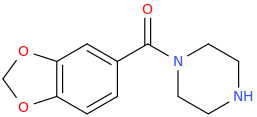
OLLIE
1-(3,4-methylenedioxy-5-methoxyphenyl)-1-piperazinylmethane
Starting with
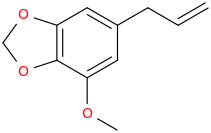
3,4-methylenedioxy-5-methoxy-1-allylbenzene, from essential oil of nutmeg
KMnO4, hot, acidic.
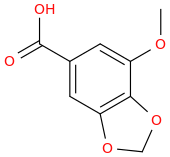
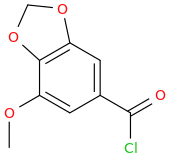
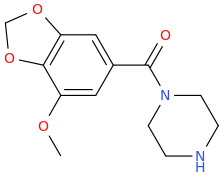
Then Wolff-Kishner the benzylic carbonyl.

OLLIE
1-(3,4-methylenedioxy-5-methoxyphenyl)-1-piperazinylmethane
Starting with

3,4-methylenedioxy-5-methoxy-1-allylbenzene, from essential oil of nutmeg
KMnO4, hot, acidic.



Then Wolff-Kishner the benzylic carbonyl.
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
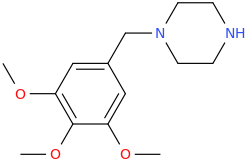
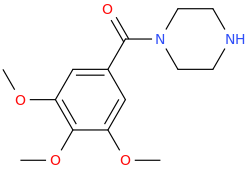
FREDERICK
1-(3,4,5-trimethoxyphenyl)-1-piperazinylmethane
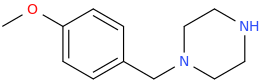
JENNIFER
1-(4-methoxyphenyl)-1-piperazinyl-methane
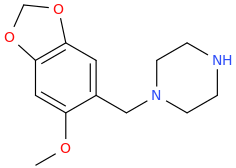
RAZ_BERRY
1-(3,4-methylenedioxy-6-methoxyphenyl)-1-piperazinylmethane
6:22 pm EST Friday Night Quitting Time.
April the 5th, 2024 Anno Domini.
Okay, just one more.
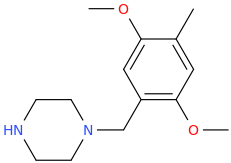
DEBORAH_DEBBIE_DEB
1-(4-methyl-2,5-dimethoxyphenyl)-1-piperazinyl-methane
Starting with
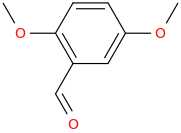
2,5-dimethoxybenzaldehyde
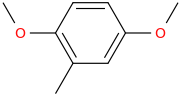
Reduction
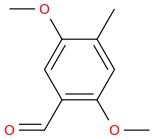
Aromatic carbonylation
Reductive Amination With Piperazine
&
Viola!
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
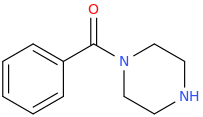
111
1-phenyl-1-piperazinylmethanone
NaBH4
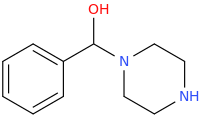
1-phenyl-1-piperazinylmethanol
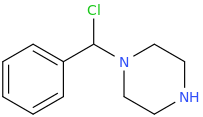
1-phenyl-1-piperazinyl-1-chloromethane
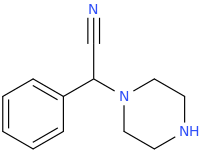
1-phenyl-1-piperazinyl-1-cyanomethane
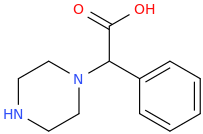
1-phenyl-1-carboxy-1-piperazinylmethane
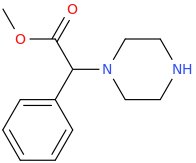
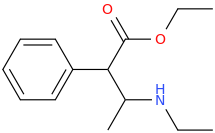
SPEEDY_CINCO
1-phenyl-1-piperazinyl-1-carbomethoxymethane
I actually think this one may be fun.
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
The Anderson Reaction:
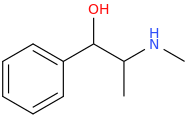
Ephedrine
1-phenyl-1-hydroxy-2-methylaminopropane
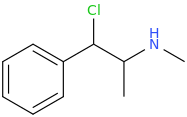
1-phenyl-1-chloro-2-methylaminopropane
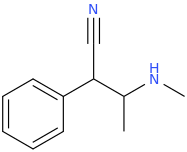
1-phenyl-1-cyano-2-methylaminopropane
Br2 / FeBr3
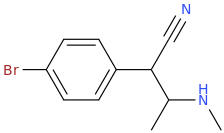
1-(4-bromophenyl)-1-cyano-2-methylaminopropane
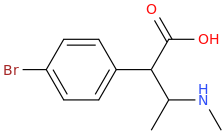
1-(4-bromophenyl)-1-carboxy-2-methylaminopropane
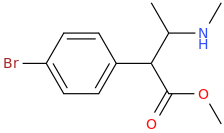
BROMO_L.O.V.E.
1-(4-bromophenyl)-1-carbmethoxy-2-methylaminopropane
In 5 Steps.

Ephedrine
1-phenyl-1-hydroxy-2-methylaminopropane

1-phenyl-1-chloro-2-methylaminopropane

1-phenyl-1-cyano-2-methylaminopropane
Br2 / FeBr3

1-(4-bromophenyl)-1-cyano-2-methylaminopropane

1-(4-bromophenyl)-1-carboxy-2-methylaminopropane

BROMO_L.O.V.E.
1-(4-bromophenyl)-1-carbmethoxy-2-methylaminopropane
In 5 Steps.
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
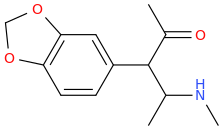
TRIS
1-(3,4-methylenedioxyphenyl)-1-acetyl-2-(methylamino)propane
Say goodbye to anhedonia and depression!
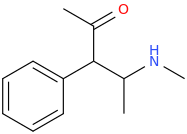
TETRAKIS
1-phenyl-1-acetyl-2-(methylamino)propane
What Do We Want? Euphoria!
Where Do We Find It? Various White Powders.
His ADHD Is Not Being Properly Treated.
TETRAKIS Can Help. Ask Your Doctor If TETRAKIS Is Right For You.
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
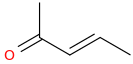
2-oxo-pent-3-ene
Next, Br2 liquid brominates the double bond on each of its two carbons
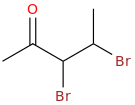
2-oxo-3,4-dibromopentane
Then, just add benzene or toluene or 1,3-benzodioxole plus Br2 and FeBr3.
Walla, 3 Productos.
I Am Not Sure What The Solvent Should Be. I'm thinking either ethyl alcohol or tetrahydrofuran.
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
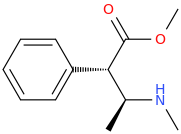
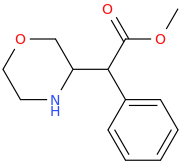
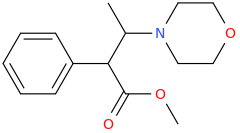
DOUBLE_DEXTER_FINN_AGAIN
(1S,2S)-1-phenyl-1-carbomethoxy-2-methylaminopropane
from
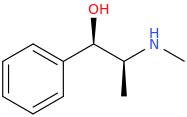
(1R,2S)-1-phenyl-1-hydroxy-2-methylaminopropane
Actually, I think the hydroxyl group may switch around the stereochemistry of the one spot after it undergoes a substitution reaction. I was never very good with the whole Khan Ingold Prelog (R,S) nomenclature.
Anyway, if that is the case, then this is the correct precursor:
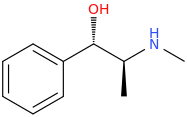
(1S,2S)-1-phenyl-1-hydroxy-2-methylaminopropane
BUT it undergoes 2 substitution reactions, so the former would be correct.
Errg, Forget Stereochemistry! All I Know Is That Ephedrine Comes In 4 Stereochemicals, And One Of Them Is Correct. Specifically, It Has A (2S).
Last edited:
Rectify
Bluelighter
- Joined
- Oct 20, 2008
- Messages
- 5,490
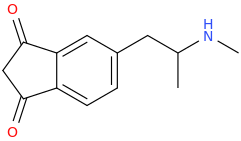
RODENTICIDE
1-(1,3-dioxoindan-5-yl)-2-methylaminopropane
This one doesn't look safe or anything. Proceed with caution, if at all.
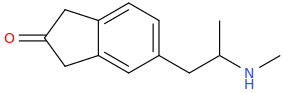
DANIEL
1-(2-oxoindan-5-yl)-2-methylaminopropane
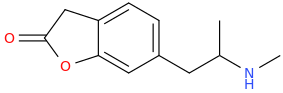
THE_LION'S_DEN
1-(2-oxo-3-oxaindan-5-yl)-2-methylaminopropane
May be metabolized into something gbl like.
Me So Horny! Lol.
Last edited: