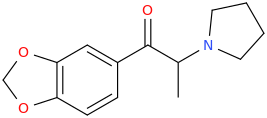
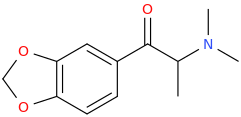
This is some weird reasoning in my opinion. Even if there is some correlation between dopamine and serotonin release in p-substituted amphetamines, it doesn't automatically mean that any analogue with modified side-chain that is still a DA releaser will be a 5-HT releaser, right?
It doesn't but is often the case.
Methcathinone > mephedrone, clephedrone, brephedrone. All have empathogenic effects. Amphetamine > 4-FA, 4-Me-amphetamine, both strong 5HT releasers. What is happening is the ring substitution is lowering DAT substrate affinity and at the same time increasing SERT substrate affinity (this is why all the substituted compounds have dosages often 10 times as much as the unsubstituted amphetamine/cathinone).
If we try the same thing with known reuptake inhibitors such as ritalin: 4-F-eph, 4-Me-eph, 4-F-mph, 4-Me-mph, 3,4-Cl-mph. All have absolutely no affinity as a SERT substrate (even though they might have affinity to simply block SERT, this however doesn't result in empathogenic effects but instead downstream antidepressant effects). Some more examples: adding a 3,4-methylenedioxy substitution to pyrovalerone doesn't confer it 5HT releasing abilities at all.
I think if something is a DAT substrate (the same way dopamine itself functions as a substrate, not some alternate binding site for example), then it has potential to be a SERT substrate with the right substitutions.
Obviously this doesn't imply that "
incorporating a 4-fluoro might increase SERT substrate affinity greatly [of phenmetrazine]" but it is pretty suggestive, in my opinion at least.